SCOPE 13 - The Global Carbon Cycle
13
|
Carbon in the Rock Cycle
|
|
S. KEMPE |
ABSTRACT
The earth's crust contains 65.5 x1021 g of C, equivalent to 0.27% of all rocks. One-quarter of this mass is non-carbonate carbon. Both forms of carbon have distinct
 13
C ‰ values: carbonate-C in the region of 0‰, on the PDB scale, and non-carbonate-C in the region of
13
C ‰ values: carbonate-C in the region of 0‰, on the PDB scale, and non-carbonate-C in the region of
 25‰ PDB. The original isotopic composition in the mantle must have been approximately
25‰ PDB. The original isotopic composition in the mantle must have been approximately
 7‰
7‰  13C PDB. The lithospheric part of the carbon cycle starts with the sedimentation of terrigeneous and planktonic material or the oceans. Consequent
subduction, metamorphism, or uplifting add the sea sediments to the continental crust. Weathering and erosion finally return the material to the oceans or continental basins. All these fluxes are generally less than 1015 g C/year. The figure for the global river transport of inorganic carbon is one of the best known in the total carbon cycle, and is 0.45 x 1015 g C/year, with evidence of increasing rates caused by human interference. Most of the weathering on the continents involves dissolution of carbonates. The carbon dioxide used for this process from soil air is recharged upon precipitation of carbonates: there is no net sink by carbonate dissolution. The only net sink of atmospheric CO2 through weathering occurs by destruction of silicates, which involves only
0.01
13C PDB. The lithospheric part of the carbon cycle starts with the sedimentation of terrigeneous and planktonic material or the oceans. Consequent
subduction, metamorphism, or uplifting add the sea sediments to the continental crust. Weathering and erosion finally return the material to the oceans or continental basins. All these fluxes are generally less than 1015 g C/year. The figure for the global river transport of inorganic carbon is one of the best known in the total carbon cycle, and is 0.45 x 1015 g C/year, with evidence of increasing rates caused by human interference. Most of the weathering on the continents involves dissolution of carbonates. The carbon dioxide used for this process from soil air is recharged upon precipitation of carbonates: there is no net sink by carbonate dissolution. The only net sink of atmospheric CO2 through weathering occurs by destruction of silicates, which involves only
0.01 0.02 x 1015
g C/year.
0.02 x 1015
g C/year.
13.1 INTRODUCTION
Models of rock cycles have been developed during the last few years (e.g. Garrels and Mackenzie, 1971, 1972; Garrels and Perry, 1974; Garrels et al., 1975). The main features of the cycles are:
(i) sedimentation of carbonates and organic matter in oceans and in continental basins, (ii) subduction and uplift of the oceanic crust, and (iii) weathering and erosion of rocks exposed on the continents.
13.2 ROCK MASSES AND THEIR CARBON CONTENT
Table 13.1 compiles estimates for the masses of different rock reservoirs of earth, while
Table 13.2 gives average composition of various rock types. The composition of the average igneous rock can also be taken as representative of the whole crust
(Garrels and Mackenzie, 1972). Carbon analyses (Fuex and Baker, 1973) of granitic rocks (14 samples) yielded 450 ppm carbonate-C and 65 ppm non-carbonate-C, while unaltered basalts and peridotites (10 samples) yielded
0-620 ppm carbonate-C(an average of 106 ppm) and 49-460 ppm non-carbonate-C (an average of 101
ppm) (14 samples). In altered basalts the carbonate-C was much larger, between 0.23 and 2%.
Table 13.1a Masses of earth in 1024 g (Garrels and Mackenzie, 1971, condensed from Table 1.4 and Figure
2.5. Reproduced by permission of W. W. Norton & Co., Inc., and by permission of the authors)
|
|
|
|
|
|
|
|
|
|
|
|
|
|
(thickness: oceans 6, continents 40, | |
|
|
|
|
|
|
|
(oceans: 268, submarine continental | |
|
|
|
margin 93, continental mountainous | |
|
|
|
regions 42, continental interiors | |
|
|
|
|
|
|
| of this, sediments and volcanics |
|
|
|
|
Table 13.1b Sediment masses in 1024 g (altered after Garrels and Mackenzie, 1971, Table 3.4. Reproduced by permission of W. W. Norton & Co., Inc., and by permission of the authors)
|
|
Limestone |
Shale |
Sandstone |
Volcanics |
Total |
|
| Total |
0.35 |
1.73 |
0.26 |
0.86 |
3.2 |
|
percentage |
11 |
54 |
8 |
27 |
100 |
|
percentage (without |
15 |
74 |
11 |
|
|
|
volcanics) |
|
|
|
|
|
| Phanerozoic |
0.26 |
0.86 |
0.20 |
0.48 |
1.80 |
|
percentage |
14 |
48 |
11 |
27 |
100 |
|
percentage (without |
20 |
65 |
15 |
|
|
|
volcanics) |
|
|
|
|
|
| Precambrian |
0.09 |
0.87 |
0.06 |
0.38 |
1.40 |
|
percentage |
7 |
62 |
4 |
27 |
100 |
|
percentage (without |
8 |
86 |
6 |
|
|
|
volcanics) |
|
|
|
|
|
|
The ratio of limestones, shales, and sandstones for post-Precambrian rocks is 20:65:15 and for Precambrian rocks 8:86:6; with a weight ratio of 1.3 between both compartments (excluding volcanic rocks), the ratio becomes 15:74:11 for all sedimentary rocks
(Garrels and Mackenzie, 1971). In the steady-state sediment cycle model of Garrels and Mackenzie (1972) (see also
Section 13.7), the ratio calculated from a river flux with the composition and quantity of present river loads is about 15:70:15, which is in accordance with the aforementioned values.
Table 13.1c Estimates of mass
and lithologic properties of major sedimentary units* (Garrels and Mackenzie,
1971, Table 8.1. Reproduced by permission of W. W. Norton & Co., Inc., and
by permission of the authors)
|
|
Sedimentary unit |
|
|
|
|
|
|
Mobile belt- |
|
|
|
Continent- |
continental |
|
|
|
shield |
shelf |
Hemipelagic |
Pelagic |
|
|
Volume (units of 106 km3) | |
127.0
|
|
|
|
|
|
241.0
|
|
|
|
|
|
|
|
|
|
|
|
|
Solid phase mass (units of 1020
g) | |
|
|
|
|
|
|
|
|
|
Liquid phase mass (units of 1020 g) | |
|
|
|
|
|
|
1187.0
|
|
| Total mass (units of 1020 g) |
|
|
|
|
|
|
|
|
| Sedimentary proportions (%) |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
2.0
|
|
|
|
|
|
|
|
|
|
3.0
|
|
|
|
|
|
|
|
|
|
|
|
|
|
The total mass of sediments is estimated according to mean depths of sediments in oceans (between 1 and 2 km) and continents (around 2.7 km), mean porosity, and mean density. This total sediment mass is estimated by Garrels and Mackenzie (1971)
(Table 13.1) to be 3.2 x 1024 g with volcanic rocks, or 2.34 x 1024 g without, while Li (1972) and Ronov and Yaroshevsky (1967) obtained a mass of 2.4 x 1024 g; Garrels and Mackenzie (1972) and Garrels and Perry (1974) assume 2.5 x 1024 g, which should include metamorphic rocks from sedimentary educts.
It is estimated that sediments contain 0.5% organic carbon by weight
(Garrels and Perry, 1974). The figure for Precambrian rocks is lower: 0.3%. Modern sediments contain 0.8 to 0.9% by weight
(Ronov, 1971).
With these figures at hand it is possible to take an inventory of carbon
(Table 13.3, Figure 13.1): assuming a basaltic composition for the oceanic crust, and a mean thickness of 4 km (6 km average oceanic crust thickness, minus 2 km sediment cover), and a density of 3 g/cm3, we obtain 268 x 106 km2 x 4 km x 3 x 1015 g/km3 = 3.22 x 1024 g; of this 106 ppm is carbonate-C = 0.34 x 1021 g, and 101 ppm non-carbonate-C = 0.32 x
1021 g.
Large differences in carbon content occur between altered and unaltered basalts. Alteration is due to local degassing processes which conduct volcanic gases into the crust. It is, however, very difficult to assess the amount of altered basalts, which could lead to an estimation of -the flux in the gaseous phase. This problem should be given more consideration and more carbon analyses are needed.
Table 13.2 Chemical analyses of `average rock' (in wt. %)
(Garrels and Mackenzie, 1971, Table 9.1. Reproduced by permission of W. W. Norton & Co., Inc., and by permission of the authors)
|
|
A |
B |
C |
D |
E |
|
Average |
Average |
Average |
Average |
Average |
|
igneous rock |
limestone |
shale |
sandstone |
sedimentary |
|
(after Brotzen, |
(after Clarke, |
(after Clarke, |
(after Pettijohn, |
rock |
|
1966) |
1924) |
1924) |
1963) |
(authors) |
|
|
|
|
|
|
|
|
SiO2 |
63.5 |
|
5.19 |
|
61.90 |
|
78.0 |
|
59.7 |
|
|
Al2O3 |
15.9 |
|
0.81 |
|
16.90 |
|
7.2 |
|
14.6 |
|
|
Fe2O3 |
2.9 |
|
0.54 |
|
4.20 |
|
1.7 |
|
3.5 |
|
|
|
|
|
|
|
|
|
|
|
2.6 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
K2O |
3.3 |
|
0.33 |
|
3.70 |
|
1.3 |
|
3.2 |
|
|
CO2 |

|
|
41.90 |
|
1.54 |
|
2.6 |
|
4.7 |
|
|
H2O (1 10 °C) |

|
|
0.21 |
|
3.90 |
|
2.2 |
|
3.4 |
|
|
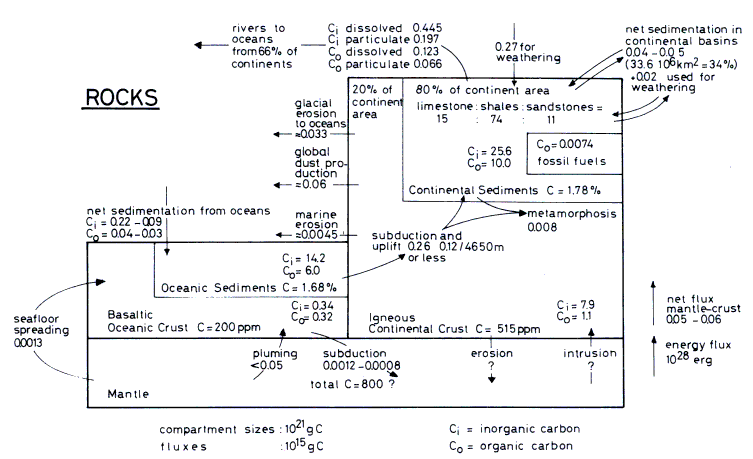
Figure 13.1 The unbalanced rock
cycle, compartment sizes, and flux rates as given in the text
Table 13.3 Masses of carbon in crustal
compartments
|
|
|
Total |
Total |
Carbon- |
Non-Carbon- |
|
|
rock |
C |
ate C |
ate C |
|
1024 g |
1021 g |
1021 g |
1021 g |
|
|
|
|
0.66 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
continental granitic crust | |
17.58 |
9.05 |
|
|
|
|
2.0 |
|
|
|
|
|
|
|
|
|
|
|
|
|
0.27 |
|
|
|
In the following, a mean carbon content of 200 ppm C for the oceanic basaltic crust is assumed. Oceanic sediments have a given composition as outlined in
Table 13.1c, with a mean CO2 content of 4.36% or 1.18% carbonate-C. The total mass of carbon
(Table 13.3) can be calculated from the corrected density and porosity as given in
Table 13.1 c) for the hemipelagic and pelagic sediments. It is not specified in the quoted reference whether the analyses
(Tables 13.1c and 13.2) include organic carbon. If it is included, the carbonate concentrations in marine sediments would be too low. By multiplying the total mass by 0.5% organic carbon, we obtain 6 x 1021 g of organic carbon in oceanic sediments.
By subtracting total sediments and basaltic oceanic crust from the total crustal mass, we obtain the continental igneous mass. This mass (17.58 x 1024 g) underlies the continents and their margins. With 450 ppm carbonate and 65 ppm
non-carbonate carbon, the carbon content of igneous masses has been calculated as in
Table 13.3.
The carbon content of the continental sediments can be calculated by taking the average sedimentary rock composition
(Table 13.2) = 4.7% CO2 = 1.28% C, multiplied by the mass of continental sediments. Similarly, organic carbon can be calculated by multiplying continental sediment mass by 0.5%.
The total carbon inventory in the crust amounts to almost 65.5 x 1021 g, of which 73% is carbonate carbon, while the rest is principally organic carbon.
Estimates of carbon in rocks have been collected from various sources: Garrels and Mackenzie (1971, p. 250) estimated the total CO2 in sediments neutralized by HCl to be 210 x 1021 g CO2 = 57 x 1021 g C. This is more than the calculated yields for all sediments. Garrels
et al. (1975, Fig. 28), and Garrels and Perry (1974, Table III) estimated the carbon mass as 5 x 1021 moles of CO2 = 60 x
1021 g carbonate-C and 1 x 1021 moles= 12 x 1021 g organic-C of which ~1%
is fossil fuel. Baes et al. (1976) calculated 30 x 1021 g carbonate-C down to a 4-km depth limit and 6.6 x 1021 g non-carbonate-C,
of which 0.01 x 1021 g or 0.15% are fossil fuels. Reiners (1973, Fig. 1) quoted Bolin (1970), with only 20 x 102 ` g of total carbon in sediments plus 0.01 x 1021 g of fossil fuel carbon.
13.3 FORMS OF CARBON IN ROCKS
Carbon has nine possible oxidation stages, i.e. from +4 to  4. Most common is the +4 in CO2 or CO2-3 (H2 CO3, or
HCO3
4. Most common is the +4 in CO2 or CO2-3 (H2 CO3, or
HCO3 ). CO (+2) plays a role as a volcanic gas and in bacterial activity. Neutral carbon is present in the minerals graphite and diamond. Most non-carbonate-C is fixed as
CnH2nOn by photosynthesis. With the help of three adenosinetriphosphate and
of two reduced pyridine nucleotides, five electrons reduce the carbon from the +4 stage to the
). CO (+2) plays a role as a volcanic gas and in bacterial activity. Neutral carbon is present in the minerals graphite and diamond. Most non-carbonate-C is fixed as
CnH2nOn by photosynthesis. With the help of three adenosinetriphosphate and
of two reduced pyridine nucleotides, five electrons reduce the carbon from the +4 stage to the
 1 level.
1 level.
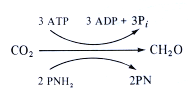
Further reduction by certain bacteria or by inorganic processes can produce CH4 (methane) as the most reduced form of carbon to occur.
13.3.1 Carbonates
Seventy-three per cent (Baes et al., 1976: 82%) of the crust-carbon is present as carbonates. Calcite (CaCO3, Ca
9-fold-coordinated, trigonal
scalenoedric, is capable of incorporating several % Mg into its lattice) and aragonite (CaCO3, Ca
6-fold-coordinated, orthorhombic
dipyramidal, is capable of taking up several % Sr into its lattice) are the dominant biogenic carbonates. Inorganically, calcite precipitates from solutions with Ca/Mg ratios larger than 1, if CO2 pressure is
favourable. With smaller Ca/Mg values, first Mg-calcite (up to 40 mol % Mg in solid solution), and then aragonite are precipitated. In very Mg-rich solutions,
monohydrocalcite, hydromagnesite, and nesquehonite can form. Dolomite (CaMg(CO3)2, trigonal
rhombohedric)
is the third carbonate mineral of wide importance; it forms by early diagenetic disintegration of high-Mg-calcites. The same holds true for the rarer magnesite (MgCO3) and huntite (CaMg3
CO3)4
). The Ca/Mg ratio has to be lower than 0.13 for the formation of dolomite, conditions normally found only in evaporative areas. Siderite (FeCO3), an iron ore, is formed by metasomatic alteration of (CaCO3, minerals, as well as in the absence of oxygen.
Figure 13.2 shows the percentage in volume of sedimentary rock as calculated for different periods of the earth's history. With increasing sediment depth and continental growth, volcanic rocks diminish and shales
(lutites) dominate. The changing proportions of limestone and dolomite should also be noted. Magnesium today is either absorbed by clay minerals or deposited as high-Mg-calcite in shallow seas, while carbonate combines preferentially with calcium to form
limestones. One of the reasons for this change is the fact that granitic continental crust tends to become more acidic. The weathering of feldspars dominates over the weathering of the more basic Mg-containing hornblendes and pyroxenes, which causes a five times larger Ca output to the rivers than that of Mg. Since the domination of land by plants in early
-Phanerozoic times, more CO2 has become available in soil, stimulating calcification in the oceans. Once the Mg/Ca ratio in the oceans (now about 5) has been lowered, calcite can be precipitated, both inorganically and organically, to form
limestones.

Figure 13.2 Volume percentage of sedimentary rocks as a function of age.
(Garrels and Mackenzie, 1971, Fig. 10.3. Reproduced by permission of Elsevier Scientific Publishing Co. and by permission of the authors.)
The isotopic composition of carbonates has changed only slightly during the earth's history
(Figure 13.3) Veizer and Hoefs (1976) have collected almost 2000
 13 C analyses of limestones and dolomites. Permian carbonates have large
13 C analyses of limestones and dolomites. Permian carbonates have large
 13C values. A decrease of 3‰ with increasing age occurred during Precambrian, as well as
-Phanerozoic times. At the Precambrian -Phanerozoic boundary, a distinct 3‰ decrease seems to have taken place. As the
13C values. A decrease of 3‰ with increasing age occurred during Precambrian, as well as
-Phanerozoic times. At the Precambrian -Phanerozoic boundary, a distinct 3‰ decrease seems to have taken place. As the
 18O values in carbonates diminish considerably with increasing age, the carbonates are, in general, heavier in
18O values in carbonates diminish considerably with increasing age, the carbonates are, in general, heavier in
 13C at a certain
13C at a certain
 18O level in Precambrian times than in later ages.
18O level in Precambrian times than in later ages.
Figure 13.4 depicts  13C values from different rocks, both carbonate
13C values from different rocks, both carbonate
 13C and the respective non-carbonate
13C and the respective non-carbonate
 13C figures. Differences in range between carbonates and igneous rocks are small compared to the difference with the non-carbonate
carbon, with values between
13C figures. Differences in range between carbonates and igneous rocks are small compared to the difference with the non-carbonate
carbon, with values between  30 and
30 and
 20
20  13C. By balancing sedimentary organic carbon with sedimentary carbonate carbon, Fuex and Baker (1973) deduce a mantle
13C. By balancing sedimentary organic carbon with sedimentary carbonate carbon, Fuex and Baker (1973) deduce a mantle
 13C of about
13C of about
 7‰.
7‰.

Figure 13.3 Histogram of  13C for limestone and dolomites from various age groups.
(Veizer and Hoefs, 1976, Fig. 5. Reproduced by permission of Pergamon Press Ltd., Oxford.)
13C for limestone and dolomites from various age groups.
(Veizer and Hoefs, 1976, Fig. 5. Reproduced by permission of Pergamon Press Ltd., Oxford.)
13.3.2 Organic Carbon
Organic carbon differs from carbonate carbon not only in isotopic composition and oxidation number, but also in the multiplicity of its chemistry. An increasing number of papers have been published in the last few years, dealing with the biogeochemistry of fossil organic carbon compounds.
Bacterial destruction and chemical diagenesis account for at first a rapid and then a slower disintegration of the entrapped organic sediment particles. In
the course of these processes, the metabolites which form and collect are very
different from their parent substances. Crude oil, natural gas, and coal are
some of the products now essential to our civilization.

Figure 13.4 Isotopic composition of carbon in different rocks. The composition of mantle carbon
( 7‰PDB) is based on estimates on the isotopic composition and relative amounts of the main crustal carbon reservoirs, and on the assumption of mantle degassing as the exclusive source of crustal carbon.
(Fuex and Baker, 1973, Fig. 1. Reproduced by permission of Pergamon Press Ltd., Oxford.)
7‰PDB) is based on estimates on the isotopic composition and relative amounts of the main crustal carbon reservoirs, and on the assumption of mantle degassing as the exclusive source of crustal carbon.
(Fuex and Baker, 1973, Fig. 1. Reproduced by permission of Pergamon Press Ltd., Oxford.)
Alkanes occur in plants and sediments, often with a predominance of odd over even chain length. In carbonates and evaporite deposits, predominance of even over odd can occur, possibly due to dehydration and reduction of lipid acids and to
alkaline evaporative environments (Welte and Waples, 1973). The
isoprenoids, phytane and pristane, are present in almost all sediments.
Amino acids and sugars, basic components of all organic tissue, can be identified in sediments. Racemization of biogenic laevo amino acids into their dextro forms can be used as a geochronological tool.
Other classifications of organic matter are based on various extraction fractions (such as humic matter). Bituminous matter is generally called
kerogen. Jackson and Moore (1976) have analysed rocks of various composition and of various ages for special kerogen properties and C, N, and P contents. C and N contents decreased in Phanerozoic times with increasing age, while the C content of cherts in prePhanerozoic times increased again. In general, the maturation process tends to remove the aliphatic and nitrogen-bearing compounds, causing an enrichment of condensed aromatic components. This leads to an inverse relation between total organic carbon and the ratio of aromatic to aliphatic kerogen carbon. The high organic carbon levels in the older cherts are interpreted as relics of the once higher CO2 level in the primordial atmosphere, while the increase in Phanerozoic times is due to:
(i) the increased sedimentation because of increased primary productivity; (ii) the increased reductivity in sediments; (iii) the change in the input pattern due to evolution (e.g., the appearance of faecal pellets from developing animals); and (iv) the longer time in maturation.
Kerogen is considered to be quite resistant to migration in the rocks, while lighter fractions, such as the
alkanes, are considered as contaminations of younger meterial, especially in
pre-Phanerozoic rocks (Smith et al., 1970).
Apart from the large isotopic difference of organic matter from carbonates, the
 13C values increase with increasing intensity of metamorphosis
(Hoefs and Frey, 1976), from
13C values increase with increasing intensity of metamorphosis
(Hoefs and Frey, 1976), from  25‰ in the unmetamorphic and anchimetamorphic rocks to
25‰ in the unmetamorphic and anchimetamorphic rocks to
 10‰ in the amphibolite facies (heated up to about 500
°C).
10‰ in the amphibolite facies (heated up to about 500
°C).
13.4 SEDIMENTATION RATES IN OCEANS
13.4.1 Carbonates
Several approaches can be applied to obtain sedimentation rates
(Broecker, 1974):
(1) By knowing the carbon difference between surface water and deep water, the mean depth of the deep water and the upwelling rate (200 cm/year calculated by
14C deficiency in the deep water), the rate can be calculated to be 2.5 g/cm2 in a thousand years, of which 2.0 g are redissolved and 0.5 g is deposited on 20% of the oceanic bottom area protruding above the
lysocline. In these areas the rate should then be 2.5 g/cm2 per thousand years
(Figure 13.5a).
(2) One can measure the carbonate content in dated cores. For those cores, taken from the 20% of areas with calcareous sediments, mean sedimentation rates of
0.9 0.8 g CaCO3/cm2 per thousand years have been established by this method. Distributed over the total ocean area, we obtain
0.2
0.8 g CaCO3/cm2 per thousand years have been established by this method. Distributed over the total ocean area, we obtain
0.2 0.16 g CaCO3 /cm2 per thousand years
(Fig. 13.5b).
0.16 g CaCO3 /cm2 per thousand years
(Fig. 13.5b).
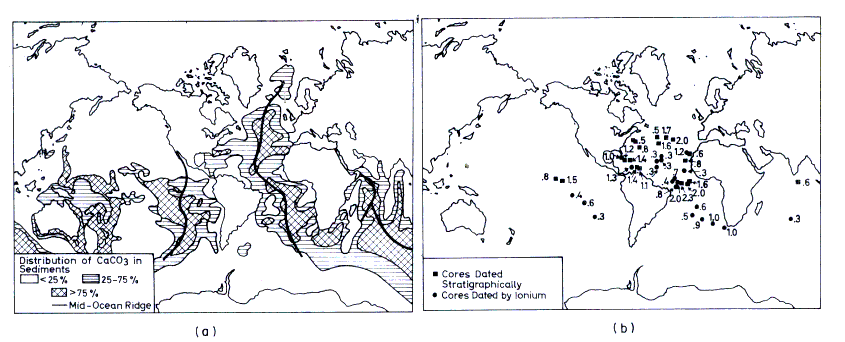
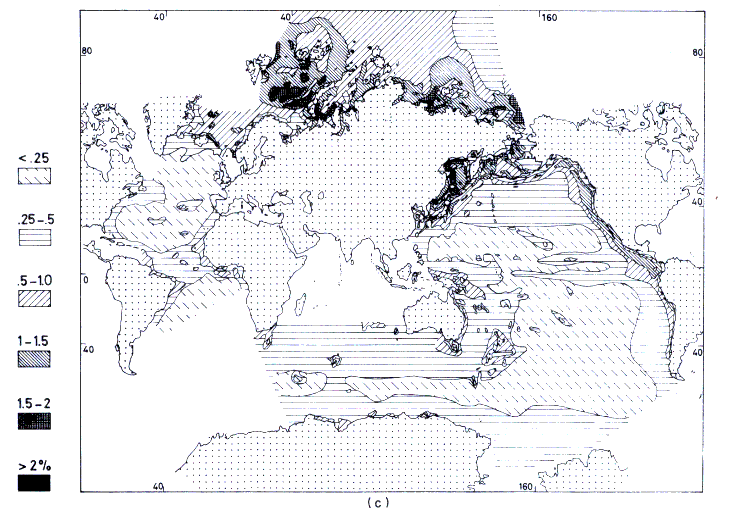
Figure 13.5 (a) Distribution of carbonate-rich oceanic sediments. (The Atlantic and Indian distributions are based on data obtained by Pierre
Biscaye, K. Venkatarathnam, James Gardner, and Thomas Kellogg, all of Lamont-Doherty Geological Observatory, Karl
Turekian, Yale University, and David Ellis, Oregon State University. The Pacific distribution is based on data compiled by A. P. Lisitzin from the
U.S.S.R.). (Broeker, 1974, Fig. 2.2. From
Chemical Oceanography by Wallace S. Broecker (© 1974 by Harcourt Brace Jovanovich, Inc. and reproduced with their permission)
(b) Actual rates of carbonate sedimentation (g/cm2 103 year).
(Broeker, 1974, Fig. 3.12. From
Chemical Oceanography by Wallace S. Broecker (© 1974 by Harcourt Brace Jovanovich, Inc. and reproduced with their permission)
(3) The sedimentation rate of carbonates should, if the system is at steady state, be equal to the Ca runoff from the continents. With an average dissolved load of 15
ppm Ca, and a stream discharge of 0.376 x 1020 g/year, we obtain a dissolved Ca flux of 0.564 x 1015 g/year, giving a minimum sedimentation of 1.41 x 1015 g CaCO3/year or 0.169 x 1015 g C/year. Divided by the area of the ocean (362 x 106
km2), the sedimentation rate is 0.39 g CaCO3 /cm2 per thousand years.
This third value is intermediate to the two figures calculated above. It does not take into account, however, the CaCO3 brought in by suspension, nor does it take rapid sedimentation at river mouths and on continental shelfs into consideration.
Broecker, for instance, calculates the percentage of detrital aluminosilicates reaching the open ocean as 6%.
By looking at these calculations, the average CaCO3 sedimentation rate seems to stay between 0.5 and 0.2 g/cm2 per thousand years. This gives us a carbonate carbon flux of
0.22 0.09 x 1015 g/year to the sediments. This is a net flux. Certainly, more carbonate is
sedimentated, which in turn is redissolved from the sediment interface.
0.09 x 1015 g/year to the sediments. This is a net flux. Certainly, more carbonate is
sedimentated, which in turn is redissolved from the sediment interface.
13.4.2 Organic Carbon
The sedimentation of organic carbon in the oceans (Figure
13.5c) is a complex process; in coastal areas, not only does continental runoff determine the amount of terrigenous plant matter introduced, but the kind, time, and amount of nutrients delivered also have a pronounced influence on the planktonic fixation of organic carbon. In the open ocean, planktonic production prevails, especially in upwelling zones and in deeper and nutrient-rich waters. In the Arctic zone, the plankton bloom occurs once during summer, while temperate climates sustain two blooms, a larger one in spring and a lesser one in autumn. In the equatorial seas, there is increased phytoplankton productivity during winter.
Suspension concentrations are higher in shelf areas; Lisitzin (1972) recorded 2-3 mg/1 as an average and 1.5 mg/l in the open ocean. An overall mean, both vertically and laterally, of one mg/1 was estimated, resulting in a global suspension mass of 1.37 x 1018 g. Half of this is stored in the Pacific and 16 x 1015 is annually exchanged by currents between the oceans.
Of this vast amount of particulate matter in the oceans, only part is carbon, and of this only very small fractions reach the ocean bottom and eventually stay there permanently (see Chapters 10 and 11, this volume). Garrels and Perry (1974) calculated the organic carbon flux by dividing the product of the total sediment mass (25000 x 1021 g) and an average organic carbon content of 0.5% by the
product of the total annual sediment flux of 6.1 x 1015 g/year x 0.5% organic carbon to obtain 0.03 x 1015 g/year. Seibold (1974) estimated a flux value of 0.04 x 1015 g/year.
The sedimentation potential of the shallow seas alone, however, is considerably larger. With a shelf area of 93
x 106 km2 (Table 13.1a) an assumed average sedimentation rate of 0.05 cm/year, a porosity of 50%, a density of 2.3, and a carbon content of 3-4% by weight, an annual 0.3-0.4 x 1015 g C/year may be deposited (this volume, Group III, Report). In fact, eutrophication of coastal areas may be instrumental in the fixation of carbon and account for some of the missing industrial CO2. The rates at which nitrogen and phosphorus are fixed and mined are 6 x 1012 and 0.4 x 1012 moles/year respectively. With a sediment C:N:P ratio of 150:15:1, eventually 60 x 1012 moles or 0.72 x 1015 g of carbon may be fixed. This amount, incidentally, is equal to the missing industrial carbon not picked up by the ocean (1.6 x 1015 g) and not remaining in the atmosphere (2.4 x
1015 g) (Dahlem Konferenzen, 1977, Group Report `Man and the global nitrogen cycle'). It is obvious that the calculation falls short of reproducing the reality. Most of the phosphorus will not be available in a dissolved form, but will be fixed chemically to soil and agricultural products.
13.4.3 Sedimentation in Continental Basins
Of the 148.9 x 106 km2 continents, 14.1 x 106 km2 belong to the Antarctic and 33.6 x 106 km2 are central regions without drainage to the oceans. Excluding the Antarctic, which has runoff only in the form of ice, 75% of the total continents are peripheral and 25% central regions (Baumgartner and
Reichel, 1975). Central areas are arid: 100.5 x 1018 g of H2O fall as precipitation over the peripheral areas, while only 8.2 x
1015 g fall in central regions. This is a percentage ratio of 92 to 8 (with respect to total precipitation over the continents, excluding the Antarctic).
In peripheral areas 62.8 x 1018 g are evaporated and 37.8 x 1018 g discharged to the oceans. This gives a runoff coefficient
(D/P) of 0.375 for peripheral regions. Central areas actually have no runoff, as all precipitation is eventually re-evaporated to the atmosphere,
P = E = 8.2 x 1018 g/year. This evaporation occurs partly from the surfaces of closed lakes and salt pans of central basins. By assuming a similar runoff coefficient to that in peripheral areas, the flux of water into the closed lakes should have a maximal size of 8.2 x 0.375 = 3.075 (x
1018 g). This is 8% of the peripheral discharge.
Sediment transport by way of river flux may be relatively larger than in peripheral regions, due to:
(i) the leaching of salt deposits often occurring in closed basins, and (ii) a high mechanical erosion resulting from sparse vegetation and from few, but violent, rain storms.
If we assume that less discharge is cancelled out by more erosion, the rate of erosion in continental basins may be estimated by multiplying the calculated runoff by the mean carbon loads established for peripheral areas (see
Chapter 12, this
volume, Table 12.1 b). The mean concentrations are:59.8 ppm HCO3 , 3.28 dissolved organic carbon (DOC), and 1.76 particulate organic carbon
(POC). The corresponding fluxes into continental basins are: 0.0362 x 1015
g HCO3
, 3.28 dissolved organic carbon (DOC), and 1.76 particulate organic carbon
(POC). The corresponding fluxes into continental basins are: 0.0362 x 1015
g HCO3 -C, 0.01 x 1015 DOC, and 0.0054 x 1015 g POC per year.
-C, 0.01 x 1015 DOC, and 0.0054 x 1015 g POC per year.
The global mean for mechanical to chemical erosion is 4.7 (Garrels and Mackenzie, 1971, Table 5.1). The mean carbon content of the suspended sediment was calculated to be 0.96%
(Garrels and Mackenzie, 1971, Table 4.11, recalculated). Multiplying the mean river salinity (111
ppm) by the total discharge from peripheral areas x 4.7 x 1%, we obtain a flux of 0.197 x 1015 g C suspended in the peripheral regions and (multiplying by 0.08) 0.0157 x 1015 g into central basins.
The sum for the total fluxes into continental basins is:
HCO3 C C |
0.036 |
x 1015 g/year |
| DOC |
0.01 |
x 1015 g/year |
| POC |
0.0054 |
x 1015 g/year |
| inorg. susp.C |
0.016 |
x 1015 g/year |
|
| total C |
0.067 |
1015 g /year |
The sedimentation rate is smaller, since roughly half of the HCO 3-C
is given off to the atmosphere with the formation of CaCO3, i.e.,
about 0.018 x 1015 g, and only part of the organic carbon is buried
in the continental basin sediments because of rapid oxidation. The sedimentation
rate should, therefore, be closer to 0.04
3-C
is given off to the atmosphere with the formation of CaCO3, i.e.,
about 0.018 x 1015 g, and only part of the organic carbon is buried
in the continental basin sediments because of rapid oxidation. The sedimentation
rate should, therefore, be closer to 0.04 0.05 x 1015 g C/year.
0.05 x 1015 g C/year.
13.5 FLUXES BETWEEN CRUSTAL COMPARTMENTS
AND MANTLE
13.5.1 Sea-floor Spreading and Subduction of Crustal Plates
If we assume that the lithosphere has been formed by
continuous addition of mantle material, we can calculate the annual net flux of
mantle material to the crust. The oldest known rocks in the crust are 3.8 x 109
years old, and the formation of a first crust, now reworked in younger rocks,
may have started 4.5 x 109 years ago. The 24 x 1024 g of
Crustal rock should then have accumulated at a rate of 5 6 x 1015
g/year. If we assume that 0.27% of this is carbon, we obtain 0.14
6 x 1015
g/year. If we assume that 0.27% of this is carbon, we obtain 0.14 0.16 x 1015
g/year delivered from the mantle. It will be extremely difficult to assess where
this net flux occurs, as this rate is one of the minor ones encountered in the
carbon cycle.
0.16 x 1015
g/year delivered from the mantle. It will be extremely difficult to assess where
this net flux occurs, as this rate is one of the minor ones encountered in the
carbon cycle.
In determining the rate of sea-floor spreading, we are faced
with difficulties. The arithmetic mean of spreading rates as calculated by Le
Pichon (1968) is 2.3 cm/year. The length of the mid-ocean ridges is estimated to
be 72 x 103 km (Wunderlich, 1973). The thickness of the basaltic
crust is 4 km (see Section 13.1); Hess (1962) assumes a thickness of 4.7 km. The
volume gain per annum is then 6.6 km3 , or (density 3 g/cm3)
20 x 1015 g. Khan (1974) arrived at a figure of 170 km3
annually generated by sea-floor spreading. He did not state, however, how he derived this large rate, but he had obviously included in
his calculation the peridotite layer below.
It is assumed that the subduction velocity has the same value
as sea-floor spreading. This is not necessarily the case, since subduction
troughs may selectively subduce those plates which have higher velocities. The
length of the subduction troughs is in fact only 44 x 103 km, as
estimated by Livingstone (1973), and plate velocities seem to remain around 3 4
cm/year (Holmes, 1965). By taking the spreading rates as subduction rates, we
obtain 4 km3 subduced annually, with the difference of about 3 km3
lifted up in mountain belts; or by taking the plate velocities of Holmes, we
find 5.3
4
cm/year (Holmes, 1965). By taking the spreading rates as subduction rates, we
obtain 4 km3 subduced annually, with the difference of about 3 km3
lifted up in mountain belts; or by taking the plate velocities of Holmes, we
find 5.3 7 km3, which would just balance the generation by spreading.
On the other hand, the annual flux from the mantle (5
7 km3, which would just balance the generation by spreading.
On the other hand, the annual flux from the mantle (5 6 x
1015 g) would amount
to about 1.7
6 x
1015 g) would amount
to about 1.7 2 km3 , which could fill the difference between the
first subduction rate and the rate of crustal generation at the ridges. But this
may be a hasty conclusion, as there are other processes by which mantle material
can exchange with the crust.
2 km3 , which could fill the difference between the
first subduction rate and the rate of crustal generation at the ridges. But this
may be a hasty conclusion, as there are other processes by which mantle material
can exchange with the crust.
Pluming is one of these processes. The upward flux of
material over hot spots in the mantle may be of a considerable size. For
approximately 20 hot spots on earth (most of them are situated beneath oceanic
crust), Khan (1974) assumes a total flux of about 6 km3 /year.
Again, this flux occurs in the mantle and it is questionable as to how much
material enters the crust. With a density of 3 g/cm3 and a carbon
content of 200 ppm, the maximal flux may be 0.0012 x 1015 g C/year. By looking
at the huge volcano chains which have erupted over hot spots (e.g. Hawaii
chain), one may think of pluming as a significant flow of material to the crust.
Crust may not only be subduced, but also eroded by mantle
convection under continents. Also, intrusions of mantle material, a possible
source of energy and material for continental volcanism, may play a certain
role. Estimates of the magnitude of these processes are, however, difficult to
obtain.
13.5.2 Subduction of Oceanic Sediments
Oceanic sediments are derived from the continents. If their
subduction recycled their material to the mantle, the flux to the mantle would
be positive, or just balanced, and no net gain of crust would result (compare
figures presented above). If, at 44 x 103 km length, a 2-km-thick
layer of oceanic sediment is subduced at a rate of 2.3 (3 4) cm/year, we obtain
a flux rate of 2 (2.6
4) cm/year, we obtain
a flux rate of 2 (2.6 3.5) km3/year. With an average total carbon
content of 1.68% for the sediments and a mean density of 2.14 (weighted mean for
pelagic and hemipelagic sediments including dissolved mass of pore solution,
recalculated from Garrels and Mackenzie, 1971, Table
8.1), the resulting carbon
flux is 0.073 (0.095
3.5) km3/year. With an average total carbon
content of 1.68% for the sediments and a mean density of 2.14 (weighted mean for
pelagic and hemipelagic sediments including dissolved mass of pore solution,
recalculated from Garrels and Mackenzie, 1971, Table
8.1), the resulting carbon
flux is 0.073 (0.095 0.13) x 1015 g/year. Note that this is about
the size of the carbonate sedimentation of the oceans (Figure
13.1). Several
questions, however, remain when we assume a reflux of sediment material to the
continents in this manner. The sediments cannot be subduced but should be
folded, partly metamorphosed, and uplifted on to the margins of continents. But
only parts of the subduction troughs are situated at continent rims (e.g. off
western South America) and appreciable lengths are found in the open oceans. How
much of this oceanic sediment material is recycled to the surface by the
activity of volcanoes? Is the present plate structure representative for the
entire length of earth history'?
0.13) x 1015 g/year. Note that this is about
the size of the carbonate sedimentation of the oceans (Figure
13.1). Several
questions, however, remain when we assume a reflux of sediment material to the
continents in this manner. The sediments cannot be subduced but should be
folded, partly metamorphosed, and uplifted on to the margins of continents. But
only parts of the subduction troughs are situated at continent rims (e.g. off
western South America) and appreciable lengths are found in the open oceans. How
much of this oceanic sediment material is recycled to the surface by the
activity of volcanoes? Is the present plate structure representative for the
entire length of earth history'?
13.5.3 Uplifting and Metamorphosis
If we assume that, eventually, all carbon deposited in the
oceans returns to the continents, we can estimate the net uplift rate of this
process. Continents have a mean height of 850 m above sea-level, and oceans a
mean depth of 3800 m. The sedimentation rate is 0.26 0.12 x 1015 g
C/year, which is lifted annually by 4650 m. This figure does not include the
carbon in the inorganic suspended matter, which is mostly settled in
epicontinental seas, on the shelf in shallow water, or on the continental margin
in medium water depths. The figure does not include the sediments deposited in
continental basins either (see Section 13.4.3), which has a mean uplift rate of
only several hundred metres.
0.12 x 1015 g
C/year, which is lifted annually by 4650 m. This figure does not include the
carbon in the inorganic suspended matter, which is mostly settled in
epicontinental seas, on the shelf in shallow water, or on the continental margin
in medium water depths. The figure does not include the sediments deposited in
continental basins either (see Section 13.4.3), which has a mean uplift rate of
only several hundred metres.
During subduction, and in the processes associated with
uplifting, some of the sediments are buried so far below the surface that
hydrostatic pressure and increased temperature will cause metamorphosis.
In Section 13.2 different figures for total sediment mass
were quoted. Garrels and Mackenzie (1972) report 2.5 x 1024 g as the
total mass including metamorphic rocks and 2.3 x 1024 g not
including metamorphic rocks (1971). The difference of 0.16 x 1024
should thus represent the mass of metamorphic rocks from sedimentary educts. The
ratio of total sediment to metamorphic rocks is 16. One-sixteenth of the
uplifted rock undergoes metamorphism, i.e., about 0.016 0.008 x 1015 g C/year.
0.008 x 1015 g C/year.
13.6 EROSION
13.6.1 Weathering
Before water, wind, or ice can transport material to the
oceans or continental basins, physical and chemical weathering processes have to
take place. Carbon, in the form of carbonic acid, is especially responsible for
the chemical destruction of rocks. The principal mineral reactions involving
carbonic acid are:
Ca(Mg)CO3 + CO2 + H2O =
Ca 2+ (Mg2+) + 2HCO3
It should be noted that for every mole of new CO2,
one mole of rock-derived CO2 3 is transported. After reprecipitation
of the carbonate, one mole CO2 is released again.
3 is transported. After reprecipitation
of the carbonate, one mole CO2 is released again.
Mg2 SiO4 + 4CO2 + 4H2O
= 2Mg2+ + 4HCO3- + H4 SiO4
is the equation expressing the weathering of a non-aluminous
silicate (here the Mg-olivine forsterite). It should be noted that of 4 moles CO2
used for weathering, only 2 moles will return to the atmosphere when carbonate
precipitates.
| 2NaAlSi3 O8 + 2CO2
+ 11 H2O |
|
= Al2 Sit Os (OH)4
+ 2Na+ + 2HCO 3 + 4H4 SiO4 3 + 4H4 SiO4 |
This equation illustrates the alteration of a feldspar (here
albite) into the residual clay mineral kaolinite. The solution becomes alkaline,
and carbonates will only precipitate if Na2(HCO3)2
is produced by strong evaporation.
If anorthite (CaAl2Si2O8) is
weathered together with 2CO2 and 3H2O, no dissolved
silica, but only kaolinite, 1 mole Ca and 2 moles HCO 3 are formed.
Upon precipitation of calcite, only half of the CO2 is returned to
the atmosphere.
3 are formed.
Upon precipitation of calcite, only half of the CO2 is returned to
the atmosphere.
Weathering of silicate minerals requires CO2 and
thus is an effective mechanism for the removal of CO2 from the air.
13.6.2 Erosion Rates
Erosion rates can be calculated in two ways: the first is to
collect as much river data as possible to estimate world erosion rate; the
second is to take a well-documented area as representative and calculate from
this value the global balance.
The first procedure was carried out by Livingstone (1963) for
the chemical loads of rivers. His calculations have been revised and connected
to the present water balance data of Baumgartner and Reichel (1975) in Kempe
(Chapter 12, this volume). It has not been possible to revise the data on
dissolved organic, particulate organic and suspended inorganic matter in the
rivers. The only global calculations given for these loads are those by Garrels
and Mackenzie (1971). The following river loads can be calculated:
balance data of Baumgartner and Reichel (1975) in Kempe
(Chapter 12, this volume). It has not been possible to revise the data on
dissolved organic, particulate organic and suspended inorganic matter in the
rivers. The only global calculations given for these loads are those by Garrels
and Mackenzie (1971). The following river loads can be calculated:
HCO3 -C -C |
0.445 |
x 1015 g/year |
| DOC |
0.123 |
x 1015 g/Year |
| POC |
0.066 |
x 1015 g/year |
| inorg. susp. C |
0.197 |
x 1015 g/year |
|
| total C |
0.831 |
x 1015 g/year |
The average SiO2 content in rivers is 13.l ppm
(Livingstone, 1963) which is derived from the weathering of silicates. The main silicate occurring in igneous rocks is plagioclase (Nao.6
2 Cao.3 8 Al1. 3 8 Si2.6 2
O8 ), a mixture of anorthite and albite, which weathers into almost equal moles of
HCO3 and SiO2 moles/2 Na+ and moles/3
Ca2+.
and SiO2 moles/2 Na+ and moles/3
Ca2+.
The total SiO2 river load amounts to (13.l x 10-6 x 37.7 x 1018 g H2O) 0.49 x 1015 g or 8.2 x 1012 moles. 8.2 x 1012 moles carbon is 0.l x 1015 g C, so about one-quarter of the
HCO3 -C in the rivers stems from the weathering of silicates. Of
this, one-third is balanced by Ca and can form carbonate, i.e., one-sixth of 0.1 x 1015 g returns to the atmosphere, one-sixth forms carbonates, which are removed from the system, i.e., annually 0.016 x 1015 g. Together with the
HCO
-C in the rivers stems from the weathering of silicates. Of
this, one-third is balanced by Ca and can form carbonate, i.e., one-sixth of 0.1 x 1015 g returns to the atmosphere, one-sixth forms carbonates, which are removed from the system, i.e., annually 0.016 x 1015 g. Together with the
HCO 3
C from the weathering of carbonates, we obtain the following balance (all units in 1015 g/year):
3
C from the weathering of carbonates, we obtain the following balance (all units in 1015 g/year):
|
|
to photosynthesis |
to sediment |
|
or to atmosphere |
remainder |
|
| derived from carbonates: |
0.34 |
0.17 |
0.17 |
|
| derived from silicates: |
0.1 |
0.016 |
0.016 |
0.07 |
|
| total: |
0.44 |
0.186 |
0.186 |
0.07 |
|
Of the rest, 0.05 x 1015 g is balanced by Na+ liberated by the silicates, while about 0.02 x 1015 g is not electrically balanced by cations and potentially increases the pH, if it is not removed by photosynthesis.
The sink for CO2,
removed from the system by silicate weathering, has the size of about
0.01 0.02 x 1015 g C/year.
0.02 x 1015 g C/year.
Another estimate on the global erosion can be drawn from the calculations done by Degens
et al. (1976, Table 4, Figure 7) for the tributary area of the Black Sea. The weighted means (sum of area times respective value/sum of area) for the total river loads into the Black Sea have been calculated
(Table 13.4). The ratio of suspended load to dissolved load is 1.7/1. A total of 240 x 1012 g stems from an area of 1.98 x 106 km2. Calculating for the total peripheral regions of the continents (100.9 x 106 km2) we obtain a global erosion rate of 12.07 x 1015 g/year. This figure should include
HCO3 C,
POC, and suspended inorganic carbon, but not DOC. By assuming a carbon content of 3%, a little larger than that for mean crustal rock (because of the large concentrations of CO2
C,
POC, and suspended inorganic carbon, but not DOC. By assuming a carbon content of 3%, a little larger than that for mean crustal rock (because of the large concentrations of CO2 3
in the water), we obtain a flux of 0.36 x 1015 g C/year from the rivers to the oceans.
3
in the water), we obtain a flux of 0.36 x 1015 g C/year from the rivers to the oceans.
Although the calculation is based on only 1% of the peripheral areas of the continents, the close agreement between this figure and the one based on the world river load is striking. The Black Sea area appears to be a particularly attractive example, because the river systems discharging into the Black Sea are derived from a wide array of geomorphologic and climatic settings
(Figure 13.6).
13.6.3 Erosion by Means Other than Running Water
The best figures available for transport by
ice, marine erosion and dust are those published by Garrels and Mackenzie (1971, Table 4.11).
The glacial flux was calculated by using an estimate by Yevteyev (1959) of 0.69
km3 of mechanical ground rock, which is annually transported by ice from the Antarctic continent. The rock is assumed to have the composition of unaltered average crustal rock. The figures calculated may be revised with newer data for the
Table 13.4 Denudation in source area of Black Sea
basin (Degens et al., 1976, Table 2. Reproduced by permission of Ecological Bulletins)

Figure 13.6 Oragraphic map of the Black Sea
drainage area. (Degens et al., 1976, Fig 1. Reproduced by permission of
Ecology Bulletins.)
ice flux from Antarctica: 1987 x 1015 g are given off to the surrounding oceans annually (Baumgartner and
Reichel, 1975). The ice flux from Greenland is 390 x 1015 g, which is 16.4% of the sum from both areas. Garrels and Mackenzie assumed that only 10% stem from areas other than the Antarctic; this proportion would give a 6% increase of the fluxes, a total flux of 1.85 x 1015 g rocks, of which 1.78% is carbon, i.e., the carbon flux with glaciers amounts to 0.033 x
1015 g C/year.
Even more difficult to obtain is the estimate of the material eroded by waves and currents along the coasts. Garrels and Mackenzie (1971) assume a flux of 0.25 x 1015 g, which would supply an additional amount of carbon of 0.0045 x 1015 g/year to the oceans.
The atmospheric dust settling over the oceans was assessed by Garrels and Mackenzie (1971) - assuming five dust storms annually
 to be 0.3/g3 cm
2 on 1% of the ocean. Annually, 0.054 x 1015 g of material settle into the oceans (average sediment content of 1.78% C assumed), which gives a carbon transport of 0.001 x 1015 g/year. Slowly settling tropospherc dust also reaches the sea. The average dust concentration in the air is 10-12
cm3 /cm3. With a density of 2.5 g/cm3, a troposphere density of 1.223 x 10-3 g/cm3, and a mass of 51.3 x
1020 g, the troposphere contains an average of 0.0105 x 1015 g of dust with a mean residence time of about one year. 0.0074 x 1015 will settle over the ocean, of which about 0.000 13 x 1015 g is carbon. The total dust flux from the continents through the atmosphere to the oceans amounts, then, to 0.0011 x 1015 g C/year.
to be 0.3/g3 cm
2 on 1% of the ocean. Annually, 0.054 x 1015 g of material settle into the oceans (average sediment content of 1.78% C assumed), which gives a carbon transport of 0.001 x 1015 g/year. Slowly settling tropospherc dust also reaches the sea. The average dust concentration in the air is 10-12
cm3 /cm3. With a density of 2.5 g/cm3, a troposphere density of 1.223 x 10-3 g/cm3, and a mass of 51.3 x
1020 g, the troposphere contains an average of 0.0105 x 1015 g of dust with a mean residence time of about one year. 0.0074 x 1015 will settle over the ocean, of which about 0.000 13 x 1015 g is carbon. The total dust flux from the continents through the atmosphere to the oceans amounts, then, to 0.0011 x 1015 g C/year.
Duce (1977) has reviewed existing data on organic carbon in aerosols
(Table 13.5). His analyses suggest that the mean organic carbon content of particles in the marine environment stays at approximately 5 pg/m3 at surface conditions and around 1.5
µg/m3 in non-urban continental areas. It is interesting to note that 80% of the carbon in ocean areas is associated with particles less than 1 µm in diameter. This implies that the POC is not derived from the sea, as experiments with air bubbles in sea-water show that the carbon ejected with sea spray is bound mainly to the larger particles. Duce suggests, therefore, that the small particles form by reactions between land-derived gaseous compounds. This would also explain the apparently large residence time of these small particles, which only seems large, as the emission of small particles is supplemented by volatile hydrocarbons, derived from plants.
Table 13.5 also gives the POC emissions, as estimated by Duce. A total of 0.056 x 1015 g is emitted annually, of which about 50% is believed to be anthropogenic and 50% natural. About 25% stems from oceanic sources and the rest is land-derived. These figures do not include inorganic carbonate carbon in dust, so that the total atmospheric particulate carbon budget seems to be at least 50 times larger than estimated by Garrels and Mackenzie (see above).
In addition to the continent-derived dust, cosmic dust also enters the atmosphere. Garrels and Mackenzie (1971) quoted Wasson et al. (1967), who presented a flux rate of
10-7 g/cm2 year; multiplied by the surface of Earth (510 x 106 km2), a total of 5.1 x 1011 g enters the atmosphere (Garrets and Mackenzie obtained 5.1 x 1012 g, which is obviously a mistake in calculation).
Table 13.5 POC in troposphere (condensed from unpublished material by Duce, 1977, by permission of the author)
|
|
|
Particulate organic carbon emission1012 g/year |
|
|
|
|
|
d > 1 µm |
d < 1 µm |
Total |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Particulate organic carbon burden in the troposphere |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
37 days |
|
|
Measurements taken from the Apollo project on the Moon gave dust
flow values of 4 x 10-9 g/cm2 year (quoted in Cailleux, 1976). The total
flux to the Moon (Moon surface 37.95 x 106 km2) is then 1.5 x 109 g/year. The surfaces and the gravitational constants of Moon and Earth relate by 0.0744 and 0.167 respectively. Both Moon and Earth collect all dust which comes across their way through space. The cross-sections of the path pipes of Moon and Earth relate to one another like the squares of the gravitational constant. Consequently, the
flux of cosmic dust to Earth must be 62 = 36 times that of the Moon. This gives a total flux of 5.465 x 1010 g/year or 1.07 x 10-8 g/cm2 year. These values are one order of magnitude lower than those by Garrels and Mackenzie.
Of all meteorites, 86% are chondrites; the remainder consists of achondrites, stony iron, and iron meteorites. The chondrites have been extensively investigated for their carbon content, which amounts to 0.04% (Gibson
et al., 1971). 2% of the chondrites are carbonaceous chondrites, which have a mean carbon concentration of 1.32% (mean of 44 analyses, Gibson
et al., 1971); Masses of meteorites and carbon content are shown in the following table:
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
14 |
|
? |
? |
|
|
totals |
100 |
5.47 x 1010g |
|
3.07 x 107 g C/year |
|
|
13.6.4 Changes in Erosion Rates
Man's activity on this planet has caused disturbances in many of the natural processes. Erosion is clearly one of them. Due to agricultural activities, suspension loads of rivers have increased. It is, however, very difficult to assess the rate of this increase.
Recently, the varve-counting technique allowed a first insight into the magnitude of man's interference. Sediment cores in the Black Sea and in Lake Van (eastern Turkey) were dated by
varve-counting. In the case of the Black Sea (Figures 13.6,
13.7) (Degens et al. 1976), sedimentation rates during Roman times remained at about 4 cm/1000 years, increasing to values twice as high in early, and thrice as high in late medieval times. Only in the last two hundred years has sedimentation slowed down again to a factor of two, principally as a consequence of soil conservation in the
U.S.S.R. In the case of Lake Van (Kempe, 1977), the sedimentation rate increased from 5 cm/ 1000 years to three times the value within the last 800 years.
Detailed records of hydrochemical parameters of rivers are available only from industrialized countries, with more regular sampling being carried out since the Second World War. Only recently have computer carbonate equilibrium models
(Kempe, 1975) made it possible to determine exact CO2 pressure curves. The parameter with the greatest influence on the
PCO2 is the pH, which has been monitored regularly in Germany, for instance, since the early 1950s.
Figure 13.8 gives an evaluation of 2000 water samples from the Elbe River at Hamburg (analyses by the Hamburger
Wasserwerke, from 1954). After computing the PCO2, monthly and annual averages were calculated for temperature and alkalinity. Both
PCO2 and alkalinity show an increase during the period sampled.
The correlation coefficient for PCO2 with time is 0.14 and for
alkalinity 0.47.

Figure 13.7 Sedimentation rate versus varve model ages
in a core from the Black Sea
(Degens et al., 1976, Fig. 2. Reproduced by permission of Ecological Bulletins.)

Figure 13.8 Alkalinity, PCO2,
temperature, and runoff of the Elbe River at Hamburg. PCO2 and annual
means calculated according to data kindly provided by the Hamburger Wasserwerke
Regression analysis yields an average alkalinity increase of
0.011 meq/l and of  log
0.003 PCO2 per year. It has not yet been established how much of
this increase can be attributed to the rise in atmospheric CO2
concentration, or to other factors such as an increased erosion through land management practices, or an enhanced respiration activity within the river due to an increased organic carbon load. Organic carbon in the Elbe has been monitored only for the last 3 years
(Figure 13.9). The trend cannot yet be appraised because of a strong seasonal variability. If, on the other hand, the decaying of organic particulate pollution is responsible for the increasing inorganic carbon load in the Elbe, we should expect a strong correlation between the discharge volume and the CO2 pressure; at low water levels the pollution tends to concentrate, thus causing a larger PCO2. No connection between these parameters can be seen in the curves of the annual averages as given in
Figure 13.8.
log
0.003 PCO2 per year. It has not yet been established how much of
this increase can be attributed to the rise in atmospheric CO2
concentration, or to other factors such as an increased erosion through land management practices, or an enhanced respiration activity within the river due to an increased organic carbon load. Organic carbon in the Elbe has been monitored only for the last 3 years
(Figure 13.9). The trend cannot yet be appraised because of a strong seasonal variability. If, on the other hand, the decaying of organic particulate pollution is responsible for the increasing inorganic carbon load in the Elbe, we should expect a strong correlation between the discharge volume and the CO2 pressure; at low water levels the pollution tends to concentrate, thus causing a larger PCO2. No connection between these parameters can be seen in the curves of the annual averages as given in
Figure 13.8.
The rivers Rhine, Danube, Inn, Elbe, Weser, and Ems in Germany were investigated for transported bulk matter
(Kempe et al., 1977) using data from the International Hydrological Decade during 1966--73
(Bundesanstalt für Gewässerkunde 1969-77). Both total suspended matter (TSM) and total dissolved matter
(TDM) show strong dependencies with runoff (Figures 13.10 and
13.11). The concentration of TSM increases with discharge volume, probably due to higher water velocities, while the concentration of TDM decreases due to a dilution effect. Total transport increases for both TSM and TDM with volume. Since Central Europe in the years
1966 73 observed a marked decline in water discharge
(Figure 13.12, compare also Figure
13.8), it is difficult to assess time-dependent trends. Partial correlation analysis reveals, however, a trend of decreasing suspension load. Calculating the correlation of TSM load with time, while keeping the runoff volume constant, the coefficient becomes
73 observed a marked decline in water discharge
(Figure 13.12, compare also Figure
13.8), it is difficult to assess time-dependent trends. Partial correlation analysis reveals, however, a trend of decreasing suspension load. Calculating the correlation of TSM load with time, while keeping the runoff volume constant, the coefficient becomes
 0.38. Considering again the large number of samples involved, this gives evidence of a sinking suspension load. The reasons for this behaviour in German rivers may be due to:
(i) the building of sewage plants, (ii) enhanced channellization to diminish bank erosion, or (iii) reforestation.
0.38. Considering again the large number of samples involved, this gives evidence of a sinking suspension load. The reasons for this behaviour in German rivers may be due to:
(i) the building of sewage plants, (ii) enhanced channellization to diminish bank erosion, or (iii) reforestation.
No data are available on the carbon content of the TSM, or of the
TDM, which is derived from evaporation residues and not from the sum of total chemical analyses.
The total erosion in Central Europe is around 0.1 mm/year and the ratio
TSM/TDM is about 0.13. The global mean of this ratio is 4.7, indicating a large surplus of suspended matter; 80% of this is discharged by South East Asian rivers alone.
13.7 MODELS OF THE SEDIMENTARY ROCK CYCLE
Garrels and Mackenzie (1972) have computed a quantitative model of the sedimentary rock cycle. The carbon fluxes of this model have been redrawn in
Figure 13.13.
The model is based on the assumption that no exchange with the crystalline parts of the crusts takes place and that the composition of the dissolved and suspended river loads has not changed significantly with time. The rock reservoir was partitioned into an older compartment, with a mean residence time of 600 million years, and a new reservoir, with a residence time of 150 million years. The total
model residence time obtained was 413 million years, which is close to estimates
given by other authors.

Figure 13.9 Dissolved plus particulate organic
carbon (TOC), Elbe River at Hamburg. Data kindly provided by the Hamburger
Wasserwerke

Figure 13.10 Suspension freight
and suspension concentration versus runoff volume from the rivers Rhine, Ems,
Weser, Elbe, Danube, and Inn, 1966 73.
(Kempe et al., 1977. Reproduced by
permission of the authors.)
73.
(Kempe et al., 1977. Reproduced by
permission of the authors.)
It is of interest that the model results in a crust
composition of 70% shales, 15% sandstones, and 15% limestone, which is close to
other estimates of the average sediment composition (see Section
13.2).
Approximately 20% of the carbon is used for weathering
silicates, which is not restored to the atmosphere by carbonate precipitation in
the ocean, but through a net surplus of oxidation over photosynthesis in the
sea. The total flux out of the oceans assumed in this way is 0.09 x 1015
g C/year. The residence time of carbonate carbon in the model is 380 million
years.
These models show that it is possible to model the complex
rock cycle with quite simple assumptions. The authors themselves stress,
however, that many questions remain unsolved and that the model does not
necessarily display the natural processes, but rather what the constructors
thought these natural processes to be. Nevertheless, they may be of great help
in understanding the relations of fluxes and compartments between different
elements and reservoirs.

Figure 13.11 Dissolved freight
and dissolved concentration versus runoff volume from the rivers Rhine, Danube,
and Inn, 1966-73. (Kempe et al., 1977. Reproduced by permission of the authors.)

Figure 13.12 Mean of
runoff volume (solid curves) for the rivers Rhine, Ems, Weser, Elbe, Danube, and
Inn, 1966-73, in comparison to their total suspended load (hatched curve) and
the mean of the runoff volume for the rivers Rhein, Donau, and Inn only in
comparison with their dissolved load. (Kempe et al., 1977. Reproduced by
permission of the authors.)

Figure 13.13 The carbon cycle
as calculated by a quantitative model of the sediment rock cycle by Garrels and
Mackenzie (1972). The model is based on a total material flux of sediments out
of the oceans (without H2O) of 6.1 x 1015 g/year, of which
3.21 x 1015 g are elements other than oxygen. Total rock mass is 2.5
x 1024 g expressed as oxides, inorganic C (Ci) given as CO2
and organic C (Co) as CH2O. Fluxes are net exchanges only
13.8 CONCLUSIONS
Most of the flux rates and pool sizes presented in this
chapter will be subject to continuous improvement with the growing body of
geochemical data. This particularly concerns data on the oceanic crust. It is
emphasized that additional carbon analyses will only cause a limited improvement
in the accuracy of the average concentration figures.
Much more data are needed on man's impact upon rates of
weathering and erosion. The interpretation of the sediment records in lakes and
coastal basins in correlation to past meteorological, hydrological, and chemical
data may reveal changes induced by man.
A comparison between the flux rates in the natural rock cycle
with those caused by the burning of fossil fuel indicates that burning of fossil
fuels will release carbon in an amount ten times that liberated by erosion. It
is for this reason that no potential sinks for the `missing carbon' in our
atmosphere presently exist in the rock cycle. It is only the long-term effect,
where rocks exercise control on atmospheric and oceanic carbon.
REFERENCES
Baes, C. F., Goeller, H. E., Olson, I. S., and Rotty, R. M.
(1976) The global carbon dioxide problem. ORNL-5194, 1-72, Oak Ridge National
Laboratory, Oak Ridge, Tennessee.
Baumgartner, A. and Reichel, E. (1975) The World Water
Balance, 1-179. R. Oldenbourg Verlag, München, Wien.
Bolin, B. (1970) The carbon cycle. Sci. Amer. 223(3),
124-132.
Broecker,W. S.(1974)ChemicalOceanography,1-124.Harcourt
Brace
Jovanovich Inc., New York, Chicago, San Francisco, Atlanta.
Bundesanstalt für Gewässerkunde and Deutsche
Forschungsgemeinschaft (eds) (1969, 1970, 1971, 1972, 1977) International
Hydrological Decade Yearbook. Fed. Rep. Germany 1966-73. Koblenz.
Cailleux, A. (1976) Géologie Générale; Terre-Lune-Planétes,
1-346. Masson, Paris, New York, Barcelona, Milan, and Les éditions Fides,
Montreal.
Dahlem Konferenzen (1977) Stumm, W. (ed.), The Global
Chemical Cycles and their Alteration by Man. Phys. Chem. Sci. Res. Rep. 2,
1-347, Abakon Verlagsges., Berlin.
Degens, E. T., Paluska, A., and Eriksson, E. (1976) Rates of
soil erosion. In: Svensson, B. H. and Söderlund, R. (eds), Nitrogen,
Phosphorus and Sulphur -Global Cycles. SCOPE Rep. 7, Ecol. Bull., 22,
185-191, Stockholm.
Duce, R. A. (1977) Speculations on the budget of particulate
and vapor phase non-methane organic carbon in the global troposphere. Pure
Appl. Geophys. 115 (in press).
Fuex, A. N. and Baker, D. R. (1973) Stable carbon isotopes in
selected granitic, mafic, and ultramafic igneous rocks. Geochim. Cosmochim.
Acta 37, 2509-2521.
Garrels, R. M. and Mackenzie, F. T. (1971) Evolution of
Sedimentary Rocks, 1-397. W. W. Norton and Comp. Inc., New York.
Garrels, R. M. and Mackenzie, F. T. (1972) A quantitative
model for the sedimentary rock cycle. Mar. Chem. 1, 27-41.
Garrels, R. M. and Perry Jr., E. A. (1974) Cycling of carbon,
sulfur and oxygen through geologic time. In: Goldberb, E. (ed.), The Sea,
vol. 5, Marine Chemistry, 303-336.
Garrels, R. M., Mackenzie, F. T., and Hunt, C. (1975) Chemical
Cycles and the Global Environment, 3rd ed., 1 -206. W. Kaufmann Inc., Los
Altos-Calif.
Gibson, E. K., Moore, C. B., and Lewis, C. F. (1971) Total nitrogen
and carbon abundances in carbonaceous chondrites. Geochim. Cosmochim. Acta 35,
599-604.
Hess, H. H. (1962) History of ocean basins. In: Cox, A. (ed.)
Plate Tectonics and Geomagnetic Reversals, 23-38. Freeman and Co., San
Francisco.
Hoefs, J. and Frey, M. (1976) The isotopic composition of
carbonaceous matter in a metamorphic profile from the Swiss Alps. Geochim.
Cosmochim. Acta 40, 945-951.
Holmes, A. (1965) Principles of Physical Geology. The
Ronald Press Co., New York.
Jackson, T. A. and Moore, C. B. (1976) Secular variations in
kerogen structure and carbon, nitrogen and phosphorous concentrations in pre-Phanerozoic
and Phanerozoic sedimentary rocks. Chem. Geol. 18, 107-136.
Kempe, S. (1975) A computer program for hydrochemical
problems in karstic water. Ann. Spéléol. 30, 699-702.
Kempe, S. (1977) Hydrographie, Warven-Chronologie and
organische Geochemie des Van Sees, Ost-Türkei. Mitt. Geol.-Paläont. Inst.
Univ. Hamburg 47, 123-208.
Kempe, S., Mycke, B., and Seeger, M. (1977) Flussfrachten
and Erosionsraten in Mitteleuropa, 1966-1973 (in preparation).
Khan, M. A. (1974) Dynamic implications of mantle hot spots. Nature
251, 596-597.
Le Pichon (1968) Sea floor spreading and continental drift.
In: Cox, A. (ed.), Plate Tectonics and Geomagnetic Reversals, 89-121.
Freeman and Co., San Francisco.
Li, Y. H. (1972) Geochemical mass balance among lithosphere,
hydrosphere and atmosphere. Am. J. Sci. 272, 119-137.
Lisitzin, A. P. (1972) Sedimentation in the world ocean. Soc Economic
Paleont. Miner. Spec. Pub. 17, 1-218, Tulsa, Oklahoma.
Livingstone, D. A. (1963) Chemical composition of rivers and
lakes. In: Fleischer, M. (ed.), Data of geochemistry, 6th ed., U.S. Geol.
Surv. Prof. Pap. 4406, 1-61.
Livingstone, D. A. (1973) The biosphere. In: Woodwell G. M.
and Pecan, E. V. (eds), Carbon and the Biosphere. AEC Symposium Series 30, 1-5, NTIS U.S. Dept. of Commerce, Springfield, Virginia.
Reiners, W. A. (1973) A summary of the world carbon cycle and
recommendations for critical research. In: Woodwell, G. M. and Pecan, E. V. (eds)
Carbon and the Biosphere. AEC Symposium Series 30, 368-382, NTIS
U.S. Dept. of Commerce, Springfield, Virginia.
Ronov, A. B. (1971) Allgemeine Entwicklungstendenzen in der
Zusammensetzung der aüßeren Erdhülle. Ber. Deutsch. Ges. Geol. Wiss. A.
Geol. Paläont. 16, 331-350.
Ronov, A. B. and Yaroshevsky, A. A. (1967) Chemical structure
of the earth crust. Geochimiya 11, 1285-1309 (in Russian).
Seibold, E. (1974) Der Meeresboden 1-183. Springer-Verlag,
Berlin, Heidelberg, New York.
Smith, I. W., Schopf, I. W., and Kaplan, I. R. (1970)
Extractable organic matter in Precambrian cherts. Geochim. Cosmochim. Acta 34,
659-675.
Veizer, I. and Hoefs, J. (1976) The nature of 18O/16O
and 13C/ 12C secular trends in sedimentary carbonate
rocks. Geochim. Cosmochim. Acta 40, 1387-1395.
Wasson, I., Adler, B. and Oeschger, H. (1967) Aluminum-26 in
Pacific sediments: implications. Science 155, 446-448.
Welte, D. H. and Waples, O. W. (1973) Über die Bevorzugung
geradzahliger n-Alkane in Sedimentgesteinen. Naturwissenschaften 60,
516-517.
Wunderlich, H.-G. (1973) Bau der Erde, I und II, 1-316.
Wissenschaftsverlag, Mannheim, Wien, Zürich.
Yevteyev, S. A. (1959) Determination of the amount of
morainal material carried by glaciers to the east Antarctic coast. Inform.
Bull. Soviet. Antarctic Exped. 11, 14-16.
|
Back to Table of Contents |
|
|
|
|
The electronic version of this publication has been
prepared at
the M S Swaminathan Research Foundation, Chennai, India.
|
![]() 13
C ‰ values: carbonate-C in the region of 0‰, on the PDB scale, and non-carbonate-C in the region of
13
C ‰ values: carbonate-C in the region of 0‰, on the PDB scale, and non-carbonate-C in the region of
![]() 25‰ PDB. The original isotopic composition in the mantle must have been approximately
25‰ PDB. The original isotopic composition in the mantle must have been approximately
![]() 7‰
7‰ ![]() 13C PDB. The lithospheric part of the carbon cycle starts with the sedimentation of terrigeneous and planktonic material or the oceans. Consequent
subduction, metamorphism, or uplifting add the sea sediments to the continental crust. Weathering and erosion finally return the material to the oceans or continental basins. All these fluxes are generally less than 1015 g C/year. The figure for the global river transport of inorganic carbon is one of the best known in the total carbon cycle, and is 0.45 x 1015 g C/year, with evidence of increasing rates caused by human interference. Most of the weathering on the continents involves dissolution of carbonates. The carbon dioxide used for this process from soil air is recharged upon precipitation of carbonates: there is no net sink by carbonate dissolution. The only net sink of atmospheric CO2 through weathering occurs by destruction of silicates, which involves only
0.01
13C PDB. The lithospheric part of the carbon cycle starts with the sedimentation of terrigeneous and planktonic material or the oceans. Consequent
subduction, metamorphism, or uplifting add the sea sediments to the continental crust. Weathering and erosion finally return the material to the oceans or continental basins. All these fluxes are generally less than 1015 g C/year. The figure for the global river transport of inorganic carbon is one of the best known in the total carbon cycle, and is 0.45 x 1015 g C/year, with evidence of increasing rates caused by human interference. Most of the weathering on the continents involves dissolution of carbonates. The carbon dioxide used for this process from soil air is recharged upon precipitation of carbonates: there is no net sink by carbonate dissolution. The only net sink of atmospheric CO2 through weathering occurs by destruction of silicates, which involves only
0.01![]() 0.02 x 1015
g C/year.
0.02 x 1015
g C/year.![]() 4. Most common is the +4 in CO2 or CO2-3 (H2 CO3, or
HCO3
4. Most common is the +4 in CO2 or CO2-3 (H2 CO3, or
HCO3![]() ). CO (+2) plays a role as a volcanic gas and in bacterial activity. Neutral carbon is present in the minerals graphite and diamond. Most non-carbonate-C is fixed as
CnH2nOn by photosynthesis. With the help of three adenosinetriphosphate and
of two reduced pyridine nucleotides, five electrons reduce the carbon from the +4 stage to the
). CO (+2) plays a role as a volcanic gas and in bacterial activity. Neutral carbon is present in the minerals graphite and diamond. Most non-carbonate-C is fixed as
CnH2nOn by photosynthesis. With the help of three adenosinetriphosphate and
of two reduced pyridine nucleotides, five electrons reduce the carbon from the +4 stage to the
![]() 1 level.
1 level.![]() 13 C analyses of limestones and dolomites. Permian carbonates have large
13 C analyses of limestones and dolomites. Permian carbonates have large
![]() 13C values. A decrease of 3‰ with increasing age occurred during Precambrian, as well as
-Phanerozoic times. At the Precambrian -Phanerozoic boundary, a distinct 3‰ decrease seems to have taken place. As the
13C values. A decrease of 3‰ with increasing age occurred during Precambrian, as well as
-Phanerozoic times. At the Precambrian -Phanerozoic boundary, a distinct 3‰ decrease seems to have taken place. As the
![]() 18O values in carbonates diminish considerably with increasing age, the carbonates are, in general, heavier in
18O values in carbonates diminish considerably with increasing age, the carbonates are, in general, heavier in
![]() 13C at a certain
13C at a certain
![]() 18O level in Precambrian times than in later ages.
18O level in Precambrian times than in later ages.![]() 13C values from different rocks, both carbonate
13C values from different rocks, both carbonate
![]() 13C and the respective non-carbonate
13C and the respective non-carbonate
![]() 13C figures. Differences in range between carbonates and igneous rocks are small compared to the difference with the non-carbonate
carbon, with values between
13C figures. Differences in range between carbonates and igneous rocks are small compared to the difference with the non-carbonate
carbon, with values between ![]() 30 and
30 and
![]() 20
20 ![]() 13C. By balancing sedimentary organic carbon with sedimentary carbonate carbon, Fuex and Baker (1973) deduce a mantle
13C. By balancing sedimentary organic carbon with sedimentary carbonate carbon, Fuex and Baker (1973) deduce a mantle
![]() 13C of about
13C of about
![]() 7‰.
7‰.![]() 13C for limestone and dolomites from various age groups.
(Veizer and Hoefs, 1976, Fig. 5. Reproduced by permission of Pergamon Press Ltd., Oxford.)
13C for limestone and dolomites from various age groups.
(Veizer and Hoefs, 1976, Fig. 5. Reproduced by permission of Pergamon Press Ltd., Oxford.)![]() 7‰PDB) is based on estimates on the isotopic composition and relative amounts of the main crustal carbon reservoirs, and on the assumption of mantle degassing as the exclusive source of crustal carbon.
(Fuex and Baker, 1973, Fig. 1. Reproduced by permission of Pergamon Press Ltd., Oxford.)
7‰PDB) is based on estimates on the isotopic composition and relative amounts of the main crustal carbon reservoirs, and on the assumption of mantle degassing as the exclusive source of crustal carbon.
(Fuex and Baker, 1973, Fig. 1. Reproduced by permission of Pergamon Press Ltd., Oxford.)![]() 13C values increase with increasing intensity of metamorphosis
(Hoefs and Frey, 1976), from
13C values increase with increasing intensity of metamorphosis
(Hoefs and Frey, 1976), from ![]() 25‰ in the unmetamorphic and anchimetamorphic rocks to
25‰ in the unmetamorphic and anchimetamorphic rocks to
![]() 10‰ in the amphibolite facies (heated up to about 500
°C).
10‰ in the amphibolite facies (heated up to about 500
°C).![]() 0.8 g CaCO3/cm2 per thousand years have been established by this method. Distributed over the total ocean area, we obtain
0.2
0.8 g CaCO3/cm2 per thousand years have been established by this method. Distributed over the total ocean area, we obtain
0.2![]() 0.16 g CaCO3 /cm2 per thousand years
(Fig. 13.5b).
0.16 g CaCO3 /cm2 per thousand years
(Fig. 13.5b).![]() 0.09 x 1015 g/year to the sediments. This is a net flux. Certainly, more carbonate is
sedimentated, which in turn is redissolved from the sediment interface.
0.09 x 1015 g/year to the sediments. This is a net flux. Certainly, more carbonate is
sedimentated, which in turn is redissolved from the sediment interface.![]() , 3.28 dissolved organic carbon (DOC), and 1.76 particulate organic carbon
(POC). The corresponding fluxes into continental basins are: 0.0362 x 1015
g HCO3
, 3.28 dissolved organic carbon (DOC), and 1.76 particulate organic carbon
(POC). The corresponding fluxes into continental basins are: 0.0362 x 1015
g HCO3![]() -C, 0.01 x 1015 DOC, and 0.0054 x 1015 g POC per year.
-C, 0.01 x 1015 DOC, and 0.0054 x 1015 g POC per year.![]() 3-C
is given off to the atmosphere with the formation of CaCO3, i.e.,
about 0.018 x 1015 g, and only part of the organic carbon is buried
in the continental basin sediments because of rapid oxidation. The sedimentation
rate should, therefore, be closer to 0.04
3-C
is given off to the atmosphere with the formation of CaCO3, i.e.,
about 0.018 x 1015 g, and only part of the organic carbon is buried
in the continental basin sediments because of rapid oxidation. The sedimentation
rate should, therefore, be closer to 0.04![]() 0.05 x 1015 g C/year.
0.05 x 1015 g C/year.![]() 6 x 1015
g/year. If we assume that 0.27% of this is carbon, we obtain 0.14
6 x 1015
g/year. If we assume that 0.27% of this is carbon, we obtain 0.14![]() 0.16 x 1015
g/year delivered from the mantle. It will be extremely difficult to assess where
this net flux occurs, as this rate is one of the minor ones encountered in the
carbon cycle.
0.16 x 1015
g/year delivered from the mantle. It will be extremely difficult to assess where
this net flux occurs, as this rate is one of the minor ones encountered in the
carbon cycle.![]() 4
cm/year (Holmes, 1965). By taking the spreading rates as subduction rates, we
obtain 4 km3 subduced annually, with the difference of about 3 km3
lifted up in mountain belts; or by taking the plate velocities of Holmes, we
find 5.3
4
cm/year (Holmes, 1965). By taking the spreading rates as subduction rates, we
obtain 4 km3 subduced annually, with the difference of about 3 km3
lifted up in mountain belts; or by taking the plate velocities of Holmes, we
find 5.3![]() 7 km3, which would just balance the generation by spreading.
On the other hand, the annual flux from the mantle (5
7 km3, which would just balance the generation by spreading.
On the other hand, the annual flux from the mantle (5![]() 6 x
1015 g) would amount
to about 1.7
6 x
1015 g) would amount
to about 1.7![]() 2 km3 , which could fill the difference between the
first subduction rate and the rate of crustal generation at the ridges. But this
may be a hasty conclusion, as there are other processes by which mantle material
can exchange with the crust.
2 km3 , which could fill the difference between the
first subduction rate and the rate of crustal generation at the ridges. But this
may be a hasty conclusion, as there are other processes by which mantle material
can exchange with the crust.![]() 4) cm/year, we obtain
a flux rate of 2 (2.6
4) cm/year, we obtain
a flux rate of 2 (2.6![]() 3.5) km3/year. With an average total carbon
content of 1.68% for the sediments and a mean density of 2.14 (weighted mean for
pelagic and hemipelagic sediments including dissolved mass of pore solution,
recalculated from Garrels and Mackenzie, 1971, Table
8.1), the resulting carbon
flux is 0.073 (0.095
3.5) km3/year. With an average total carbon
content of 1.68% for the sediments and a mean density of 2.14 (weighted mean for
pelagic and hemipelagic sediments including dissolved mass of pore solution,
recalculated from Garrels and Mackenzie, 1971, Table
8.1), the resulting carbon
flux is 0.073 (0.095![]() 0.13) x 1015 g/year. Note that this is about
the size of the carbonate sedimentation of the oceans (Figure
13.1). Several
questions, however, remain when we assume a reflux of sediment material to the
continents in this manner. The sediments cannot be subduced but should be
folded, partly metamorphosed, and uplifted on to the margins of continents. But
only parts of the subduction troughs are situated at continent rims (e.g. off
western South America) and appreciable lengths are found in the open oceans. How
much of this oceanic sediment material is recycled to the surface by the
activity of volcanoes? Is the present plate structure representative for the
entire length of earth history'?
0.13) x 1015 g/year. Note that this is about
the size of the carbonate sedimentation of the oceans (Figure
13.1). Several
questions, however, remain when we assume a reflux of sediment material to the
continents in this manner. The sediments cannot be subduced but should be
folded, partly metamorphosed, and uplifted on to the margins of continents. But
only parts of the subduction troughs are situated at continent rims (e.g. off
western South America) and appreciable lengths are found in the open oceans. How
much of this oceanic sediment material is recycled to the surface by the
activity of volcanoes? Is the present plate structure representative for the
entire length of earth history'?![]() 0.12 x 1015 g
C/year, which is lifted annually by 4650 m. This figure does not include the
carbon in the inorganic suspended matter, which is mostly settled in
epicontinental seas, on the shelf in shallow water, or on the continental margin
in medium water depths. The figure does not include the sediments deposited in
continental basins either (see Section 13.4.3), which has a mean uplift rate of
only several hundred metres.
0.12 x 1015 g
C/year, which is lifted annually by 4650 m. This figure does not include the
carbon in the inorganic suspended matter, which is mostly settled in
epicontinental seas, on the shelf in shallow water, or on the continental margin
in medium water depths. The figure does not include the sediments deposited in
continental basins either (see Section 13.4.3), which has a mean uplift rate of
only several hundred metres.![]() 0.008 x 1015 g C/year.
0.008 x 1015 g C/year.![]()
![]() 3 is transported. After reprecipitation
of the carbonate, one mole CO2 is released again.
3 is transported. After reprecipitation
of the carbonate, one mole CO2 is released again.![]() 3 are formed.
Upon precipitation of calcite, only half of the CO2 is returned to
the atmosphere.
3 are formed.
Upon precipitation of calcite, only half of the CO2 is returned to
the atmosphere.![]() balance data of Baumgartner and Reichel (1975) in Kempe
(Chapter 12, this volume). It has not been possible to revise the data on
dissolved organic, particulate organic and suspended inorganic matter in the
rivers. The only global calculations given for these loads are those by Garrels
and Mackenzie (1971). The following river loads can be calculated:
balance data of Baumgartner and Reichel (1975) in Kempe
(Chapter 12, this volume). It has not been possible to revise the data on
dissolved organic, particulate organic and suspended inorganic matter in the
rivers. The only global calculations given for these loads are those by Garrels
and Mackenzie (1971). The following river loads can be calculated:![]() and SiO2 moles/2 Na+ and moles/3
Ca2+.
and SiO2 moles/2 Na+ and moles/3
Ca2+.![]() -C in the rivers stems from the weathering of silicates. Of
this, one-third is balanced by Ca and can form carbonate, i.e., one-sixth of 0.1 x 1015 g returns to the atmosphere, one-sixth forms carbonates, which are removed from the system, i.e., annually 0.016 x 1015 g. Together with the
HCO
-C in the rivers stems from the weathering of silicates. Of
this, one-third is balanced by Ca and can form carbonate, i.e., one-sixth of 0.1 x 1015 g returns to the atmosphere, one-sixth forms carbonates, which are removed from the system, i.e., annually 0.016 x 1015 g. Together with the
HCO![]() 3
C from the weathering of carbonates, we obtain the following balance (all units in 1015 g/year):
3
C from the weathering of carbonates, we obtain the following balance (all units in 1015 g/year):![]() 0.02 x 1015 g C/year.
0.02 x 1015 g C/year.![]() C,
POC, and suspended inorganic carbon, but not DOC. By assuming a carbon content of 3%, a little larger than that for mean crustal rock (because of the large concentrations of CO2
C,
POC, and suspended inorganic carbon, but not DOC. By assuming a carbon content of 3%, a little larger than that for mean crustal rock (because of the large concentrations of CO2![]() 3
in the water), we obtain a flux of 0.36 x 1015 g C/year from the rivers to the oceans.
3
in the water), we obtain a flux of 0.36 x 1015 g C/year from the rivers to the oceans.![]() to be 0.3/g3 cm
2 on 1% of the ocean. Annually, 0.054 x 1015 g of material settle into the oceans (average sediment content of 1.78% C assumed), which gives a carbon transport of 0.001 x 1015 g/year. Slowly settling tropospherc dust also reaches the sea. The average dust concentration in the air is 10-12
cm3 /cm3. With a density of 2.5 g/cm3, a troposphere density of 1.223 x 10-3 g/cm3, and a mass of 51.3 x
1020 g, the troposphere contains an average of 0.0105 x 1015 g of dust with a mean residence time of about one year. 0.0074 x 1015 will settle over the ocean, of which about 0.000 13 x 1015 g is carbon. The total dust flux from the continents through the atmosphere to the oceans amounts, then, to 0.0011 x 1015 g C/year.
to be 0.3/g3 cm
2 on 1% of the ocean. Annually, 0.054 x 1015 g of material settle into the oceans (average sediment content of 1.78% C assumed), which gives a carbon transport of 0.001 x 1015 g/year. Slowly settling tropospherc dust also reaches the sea. The average dust concentration in the air is 10-12
cm3 /cm3. With a density of 2.5 g/cm3, a troposphere density of 1.223 x 10-3 g/cm3, and a mass of 51.3 x
1020 g, the troposphere contains an average of 0.0105 x 1015 g of dust with a mean residence time of about one year. 0.0074 x 1015 will settle over the ocean, of which about 0.000 13 x 1015 g is carbon. The total dust flux from the continents through the atmosphere to the oceans amounts, then, to 0.0011 x 1015 g C/year.
![]() log
0.003 PCO2 per year. It has not yet been established how much of
this increase can be attributed to the rise in atmospheric CO2
concentration, or to other factors such as an increased erosion through land management practices, or an enhanced respiration activity within the river due to an increased organic carbon load. Organic carbon in the Elbe has been monitored only for the last 3 years
(Figure 13.9). The trend cannot yet be appraised because of a strong seasonal variability. If, on the other hand, the decaying of organic particulate pollution is responsible for the increasing inorganic carbon load in the Elbe, we should expect a strong correlation between the discharge volume and the CO2 pressure; at low water levels the pollution tends to concentrate, thus causing a larger PCO2. No connection between these parameters can be seen in the curves of the annual averages as given in
Figure 13.8.
log
0.003 PCO2 per year. It has not yet been established how much of
this increase can be attributed to the rise in atmospheric CO2
concentration, or to other factors such as an increased erosion through land management practices, or an enhanced respiration activity within the river due to an increased organic carbon load. Organic carbon in the Elbe has been monitored only for the last 3 years
(Figure 13.9). The trend cannot yet be appraised because of a strong seasonal variability. If, on the other hand, the decaying of organic particulate pollution is responsible for the increasing inorganic carbon load in the Elbe, we should expect a strong correlation between the discharge volume and the CO2 pressure; at low water levels the pollution tends to concentrate, thus causing a larger PCO2. No connection between these parameters can be seen in the curves of the annual averages as given in
Figure 13.8.
![]() 73 observed a marked decline in water discharge
(Figure 13.12, compare also Figure
13.8), it is difficult to assess time-dependent trends. Partial correlation analysis reveals, however, a trend of decreasing suspension load. Calculating the correlation of TSM load with time, while keeping the runoff volume constant, the coefficient becomes
73 observed a marked decline in water discharge
(Figure 13.12, compare also Figure
13.8), it is difficult to assess time-dependent trends. Partial correlation analysis reveals, however, a trend of decreasing suspension load. Calculating the correlation of TSM load with time, while keeping the runoff volume constant, the coefficient becomes
![]() 0.38. Considering again the large number of samples involved, this gives evidence of a sinking suspension load. The reasons for this behaviour in German rivers may be due to:
(i) the building of sewage plants, (ii) enhanced channellization to diminish bank erosion, or (iii) reforestation.
0.38. Considering again the large number of samples involved, this gives evidence of a sinking suspension load. The reasons for this behaviour in German rivers may be due to:
(i) the building of sewage plants, (ii) enhanced channellization to diminish bank erosion, or (iii) reforestation.![]() 73.
(Kempe et al., 1977. Reproduced by
permission of the authors.)
73.
(Kempe et al., 1977. Reproduced by
permission of the authors.)