15 |
The Ocean System-Ocean / Climate and Ocean / CO2 Interactions |
|
HANS OESCHGER |
|
|
Physics Institute, University of Bern, |
|
|
Sidlerstrasse 5, CH-3012 Bern, |
|
|
Switzerland |
This chapter serves the purpose to demonstrate the achievements and the potential of studies of information on earth and planetary system processes recorded in natural archives, such as ocean and lake sediments, peat bogs, tree-rings, and polar ice. Especially valuable information carriers are the radioactive and stable isotopes of elements like C, H, and O. As an example 14C analyses, beside dating, make the reconstruction of solar modulation of cosmic radiation possible, enable estimates of the CO2 fluxes between atmosphere and ocean and of the turnover characteristics of ocean water. CO2 measurements on air occluded in polar ice enabled reconstruction of the anthropogenic CO2 increase and showed that the atmospheric CO2/air ratio varied also in the pre industrial era. Detailed studies of a series of environ- mental parameters indicate the existence of a climatic flip-flop mechanism in the North Atlantic area during the Wisconsin.
An essential point is the observation that the earth system mechanisms are so complex that for real progress in their understanding, studies making use of modern analytical methods which reveal the experiments the system performed in the past play an important role.
The central focus of the proposed International Geosphere-Biosphere Programme (IGBP) is 'to describe and understand the interactive physical, chemical and biological processes that regulate the total earth system, the unique environment for life, the changes that are occurring in this system, and the manner in which they are influenced by human actions'.
This holistic view of the environment emerged from studies of the global geophysical and biogeochemical experiments which man has been performing since about 150 years ago, but also from the reconstruction of experiments which nature itself has performed in the past. These experiments show the complex interactions between the individual parts of the environmental system on which hitherto research has tended to concentrate.
In the holistic studies, tracers as the radioactive and stable isotopes play a major role. Much of the discussion in this paper is therefore devoted to information on environmental systems and especially ocean system processes revealed from isotope studies. Lesser emphasis is given to the more classical theoretical and experimental studies which will certainly be fundamental to the understanding of the ocean system and its interactions in the future. During the last few years, due to the rising atmospheric CO2 content and its probable impact on climate, the ocean/CO2 cycle interaction and the ocean/climate interaction have received considerable attention by scientists and those interested in man's impact on the environment. In this chapter, therefore, main emphasis is given to these topics and recommendations are made regarding the improvement of knowledge on the interaction of the ocean with the other parts of the environmental system.
Among the great number of ways in which the ocean influences the environment, we emphasize the following:
-Ocean circulation helps to distribute energy over the globe.
-The oceans are the main source of water vapour which carries latent heat, condenses and falls as
precipitation, partly over the continents.
-The atmospheric CO2 concentration is determined by the partial pressure of dissolved CO2 in the ocean surface. This partial pressure in turn is determined by the chemical equilibria of the carbonate system.
-Atmospheric and oceanic circulation interact in a very complex manner. This non
linear system at present seems to oscillate between different modes of operation. An example is the quasi-periodical suppression of the upwelling of subsurface water in the Equatorial Pacific Ocean combined with a changing wind system. This so-called
El Niño phenomenon leads to strong increases in the temperature of Equatorial Pacific surface water and has a significant influence on weather and climate of a great part of the earth's surface.
-During the last glaciation the ocean showed a different circulation pattern and seems to have oscillated between two quasi-stable modes of operation.
This points to the sensitivity of the ocean circulation to changing boundary conditions, as sea-level, salinity, sea-ice cover, solar irradiation, etc.
-Related to man's impact on the environment, the ocean plays an important role since it is the main sink for CO2 emitted by human activities, and since it dampens (due to its heat capacity) changes of the energy balance at the earth's surface induced by the increase of 'greenhouse' gases. Today, the uptake of CO2 and excess heat can be estimated with some confidence. However, the open question remains if the ocean in this period of global change will operate as it did during the last decades and centuries.
As a basis for the discussion of the physical, chemical, and biological role of the ocean in a broad spectrum of processes on the earth's surface, in the following we introduce a concept of an environmental system (E.S.) research which evolved from isotope and tracer studies.
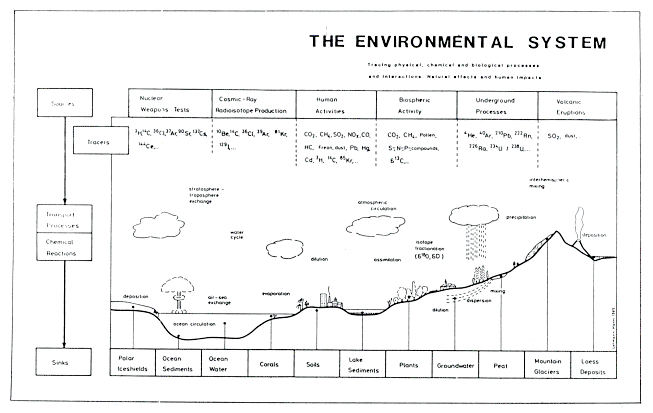
Figure 15.1 illustrates this research concept. The E.S. includes the entirety of physical, chemical, and biological processes, acting upon the earth's surface and in the atmosphere. The various parts of the system interact in various dynamic sequences and are in contact with the planetary and galactic systems. The E.S., as defined here, agrees largely with the climate system as it is generally defined, but stronger emphasis is given to chemical and biological processes. Special attention is drawn to the parameters which can be studied in natural archives and therefore enable the reconstruction of ancient system states.
The main components of the E.S. are the atmosphere, the hydrosphere, including the oceans and continental waters, the cryosphere, consisting of the polar ice sheets, sea ice, and mountain glaciers, the biosphere, consisting of the marine and continental living organisms, and the lithosphere, with bedrock and sediments which interface with the hydrosphere.
The energy of the sun is driving the dynamic processes in the E.S. It causes atmospheric circulation and oceanic mixing, and due to evaporation and precipitation, the cycling of water. The energy balance determines the climatic conditions at individual locations on the earth's surface. It is affected by scattering and reflection of short wavelength solar radiation in the atmosphere and on the earth's surface, and by the infrared radiation emission, absorption and re-emission by the surface and by water vapour and gases (CO2, O3, ...) in the atmosphere.
The dynamic cycles of some elements, such as C, N, and O, involve interactions between several of the E.S. components.
Figure15.1 Diagram of the environmental system research concept based on isotopes and tracers.
Dust particles and aerosols are injected into the atmosphere by wind action, volcanic eruptions, biospheric processes, and human activities. They reappear on the earth's surface as dry fallout or wet deposition.
Of special interest for our approach to the understanding of the E.S. processes are the radioactive and stable isotopes which constitute ideal tracers for a variety of processes and their dynamics. Radioactive isotopes have different origins:
(a) They are formed by interaction of cosmic radiation with atoms in the upper atmosphere (e.g.
10Be, 14C, 36Cl, 39Ar , 81Kr)
(b) They are also introduced to the E.S. as a result of man's use of nuclear fusion and fission (e.g.
3H, 37Ar , 85Kr)
(c) They are released from the earth's crust as products of the natural decay series of U and Th (e.g.
222Rn, 210Pb).
Since halflives vary from days (37Ar) to hundreds of thousands of years ( 10Be, 36Cl, 81Kr), information about time constants of natural processes over a very wide range is attainable. Each isotope has its characteristic field of applications which may reach far beyond that of merely dating.
Stable isotopes (e.g. 2H, 13C, 15N, 18O) are other important sources of information. Phase transitions, chemical reactions and diffusion processes produce small changes in the natural isotope ratios. They reflect the conditions at which the processes occurred. Elements originating from different natural reservoirs can often be distinguished based on their different isotopic composition. Samples of air, water, and ice, organic materials and sediments, taken from any part of the E.S., thus contain information on its static characteristics, like the partitioning of water between atmosphere, cryosphere, and ocean, but also its dynamic characteristics, like mixing and circulation, and exchange processes in and between the different system components. This and other information can also be derived from the concentration levels of chemical elements and molecules, pollen and dust. A complete set of all these parameters defines the state of the E.S. As 'fingerprint parameters' they are continuously recorded in the natural archives, as in polar ice sheets, mountain glaciers, ocean and lake sediments, and organic materials like tree-rings or peat and coral deposits. Analyses of sequential samples allow the reconstruction of the historical evolution of the E.S.
Mathematical simulation models play an important role in this research. The models are based on the fundamental physical, chemical, and biological knowledge. Since the various processes are very complex, it is often necessary to simplify the equations and to use empirical procedures to arrive at conclusions. Of course, the models should produce system responses to various perturbations which closely agree with observations. Man's impact upon the environment is an example of a present perturbation. Past system perturbations and responses are revealed from the natural archives.
The global carbon cycle
Carbon is essentially exchanged between the four reservoirs shown in Figure 15.2. Due to the burning of fossil fuels and the human impact on the biomass (deforestation and changing land use), the carbon concentrations in the atmosphere and ocean are rising. The atmosphere contains at present some 700 Gt (1 Gt = 1 Gigaton = 1012 kg) carbon in form of CO2. Precise, continuous atmospheric CO2 measurements have only been performed since 1958. Since then the CO2 concentration has increased from 315 ppm to 345 ppm in 1985. The atmospheric CO2 significantly influences the terrestrial radiation balance. It is expected that its increase will lead to a global warming.
The ocean contains roughly 40 000 Gt, about 60 times the atmospheric amount. The carbon is present in inorganic form as dissolved CO2 [CO2, aq] (~1 %), as bicarbonate [HCO-3](~90%) and carbonate ions [CO32-]( ~9%), but also as dissolved organic carbon compounds, ca. 3% of the inorganic C. The sum [CO2, aq] + [HCO-3]+ [CO32- ] is often referred to as SCO2 or total CO2.
Figure 15.2 Global carbon system main reservoirs and fluxes. Units are gigatons (109 T) C for amounts, and gigatons C per year for fluxes. Steady state assumed.
The size of the carbon pool on the terrestrial biosphere is difficult to estimate because of large regional differences in its distribution. The values range between 2000 and 3000 Gt or 3 to 4 times the amount of atmospheric CO2.
In sediments, carbon is stored in huge amounts, estimated at 60 000 000 Gt.
Of course, each of these main reservoirs represents itself as a very complicated system and may be subdivided further.
Carbon is exchanged between the different reservoirs. The exchange tries to bring the system to an equilibrium state. In the steady state in each reservoir the influxes compensate the losses.
In quasi-equilibrium states the atmospheric CO2 content is determined by pCO2, the CO2 partial pressure averaged over the ocean surface. As shown in Figure 15.3, pCO2 is significantly varying, mainly with latitude. It is determined by the chemical equilibria between the dissolved carbonate species H2CO3, HCO-3 CO32- and is essentially a function of SCO2, alkalinity, and temperature. SCO2 and alkalinity, however, are depleted in the surface ocean because biologically produced organic and carbonate particles sink to greater depth, where most of them get oxidized (organics) or redissolved (carbonates). The surface depletion of SCO2 is between 10 and 20% compared to the deep water value.
The marine biological activity depends mainly on the availability of the nutrients phosphate and nitrate. In most of the ocean surface, these nutrients are essentially consumed and at very low concentrations. This is not the case in the Pacific Equatorial Ocean due to upwelling of nutrient and CO2-rich subsurface water. This leads to a maximum in pCO2 in the water in this region (see Figure 15.3). Also, in the Southern Ocean around Antarctica the nutrients are abundant in surface waters, probably since vertical circulation or lack of light are limiting the photosynthetic activity.
Figure 15.3 North-south section of pCO2 in surface water of the Pacific Ocean. The equatorial maximum is due to upwelling of CO2-rich subsurface water. The dashed line shows the atmospheric CO2 concentration. After Broecker et al. (1980)
The essential role, which the marine biological activity plays in controlling the atmospheric CO2 concentration, is demonstrated by the estimate that an ocean without biological activity would have an average ocean surface pCO2 of between two and three times the preindustrial atmospheric CO2 concentration of 280 ppm, and that in a stagnant ocean, in which all the nutrients were consumed, pCO2 in the surface water would correspond to a an atmospheric. CO2 concentration of half the atmospheric value. Thus the complex interaction of physical, chemical and biological processes in the ocean determines such an important E.S. parameter as the atmospheric CO2 content. It is evident that changes in the oceanic behaviour lead to changes in the atmospheric CO2 concentration.
The carbon isotopic ratios in the CO2 cycle/ocean system
Natural carbon consists of the two stable isotopes 12C (~98.9%) and 13C ( ~1.1% ). The interaction of secondary cosmic ray neutrons with 14N nuclei in the atmosphere leads to the production of radioactive 14C which gets oxidized to 14CO2. In the atmosphere the 14C/12C ratio is of the order of 10-12; the halflife of 14C is 5730 yr and the specific activity of living organic matter ca. 13.5 d pm g-1 C. Figure 15.4 shows the 14C/12C ratios in the atmosphere/surface ocean/subsurface ocean system. The atmospheric 14C/12C ratio (1.00) is defined as 100% modern carbon. It is determined by production and radioactive decay of 14C in the atmosphere and its net fluxes into the oceanic and biomass carbon reservoirs. The 14C/12C ratios in the surface ocean (0.95) are determined by the net 14C flux into this reservoir from the atmosphere, the radioactive decay, and the net flux into the subsurface ocean. In the subsurface ocean ( 14C/12C = 0.84), the net 14C in-flux from the surface ocean is balanced by the radioactive decay. Based on the ratio of 0.84/0.95 for the 14C/12C ratios in the subsurface and the surface ocean, an apparent 14C age for the subsurface ocean water of ca. 1000 yr can be calculated.
A slowing down or a speeding up of ocean circulation and mixing over millenia would be reflected in changes of the apparent 14C age of subsurface water. Since about 93% of the cosmic ray produced 14C is in the deep ocean and only about 4% in the surface ocean and the atmosphere, a change in ocean dynamics would only slightly affect the 14C/12C ratio of the carbonate system in the subsurface ocean. The change would be mainly visible in shifting surface ocean and atmospheric 14C/12C ratios.
Figure 15.4 Information on CO2 and climate system, CO2 content, carbon isotopes, and their natural records.
In Figure 15.4 also the 13C/12C ratios in atmospheric CO2 and in the carbonate system of the ocean reservoirs are given. They are expressed as permil deviations (d 13C) from a standard. The mean d13C value of the ECO2 in the surface ocean is by ca. 1.5% higher than that in the subsurface ocean. This difference is due to fractionation during formation of organic matter. In aquatic plants d13C is by about 20% lower than in the ECO2 in the water in which the plants grow. Animals consuming the plants incorporate in their tissues carbon of nearly the same isotopic composition as the plants. Due to the sinking down of organic particles, carbon with somewhat lower d13C is withdrawn from the surface ocean, leaving behind ECO2 slightly enriched in 13C compared to the subsurface ocean. The difference between the mean surface and the mean subsurface d13C values in ECO2 thus is a measure of the average biological activity in the surface ocean which in turn determines pCO2 and correspondingly the atmospheric CO2 content.
The carbon isotopic ratios in the atmosphere and in the ocean are recorded in various natural archives, like polar ice, tree-rings, and sediments. In natural ice, air bubbles constitute samples of the air at the time of ice formation. From the extracted ancient air, not only can 13C/12C and 14C/12C ratios of the CO2 be measured but so too can the CO2/air ratio.
Surface dwelling planktonic foraminifera reflect the 14C/12C and 13C/12C ratios of ECO2 in surface water, and, likewise, bottom dwelling benthic foraminifera those of the bottom water. As both kinds of foraminifera are deposited in ocean sediments, parameters relevant to the carbon cycle and its dynamics cannot only be monitored in atmosphere and ocean from present into the future but can also be reconstructed from the past.
Disturbances of the carbon cycle
The release of CO2 due to human activities has led to disturbances of the atmospheric CO2 content and the isotopic ratios 13C/12C and 14C/12C in the exchanging carbon reservoirs. In addition, the 14C/12C ratio in the atmosphere, biosphere, and ocean has been considerably increased as a consequence of nuclear weapon tests. On the other hand, there are also natural changes in both the carbon contents of the reservoirs and their isotopic ratios.
The analysis of these perturbations yields clues regarding ocean circulation and mixing and their time history as well as changes in the ocean surface chemistry.
Table 15.1 shows the expected changes in one or several of the atmospheric parameters (CO2/air, 13C/12C, 14C/12C) related to the CO2 cycle due to human or natural perturbations of the carbon cycle.
The emission of fossil CO2 into the atmosphere leads to an increase of CO2/air and a decrease of 14C/12C and of 13C/12C, since in the fossil CO2 all the 14C atoms have decayed, and since the 13C/12C ratio in fossil fuels is ca. 18% lower than in the atmospheric CO2. The emission of CO2 from biomass burning also leads to a CO2 increase and a decrease of the 13C/12C ratio. The atmospheric 14C/ 12C ratio is not affected, since in biomass this ratio is close to that of the atmosphere.
Table 15.1 Carbon cycle disturbances. Expected changes of atmospheric parameters
|
pCO2 |
14C |
13C |
|
EXTERNAL FORCING |
|||
|
Cosmic radiation |
X |
||
|
Biomass destruction |
X |
X |
|
|
Fossil fuels |
X |
X |
X |
|
Nuclear tests |
X |
||
|
INTERNAL SYSTEM CHANGES |
|||
|
CO2 partitioning |
X |
X |
X |
|
Cycle dynamics |
X |
||
During atmospheric nuclear weapon tests, huge amounts of 14C were produced. After the test series in 1960 and 1961 the 14C/12C ratio in tropospheric CO2 had about doubled by 1963 (Nydal and Lovseth, 1983, and Figure 15.5). Also in the upper ocean layers, the 14C/ 12C ratio started to rise, and by detailed measurements on ECO2 in ocean water samples, the penetration of the nuclear weapon 14C pulse into the ocean could be followed (Broecker et al., 1980).
To estimate the uptake of a disturbance at the atmosphere-ocean interface often simple one-dimensional (1D) diffusive models have been used. One example is the box-diffusion (BD) model (Figure 15.6), which consists of a surface mixed layer and a diffusive subsurface ocean with constant eddy diffusivity K. The eddy diffusion in these 1D geochemical models describes all kinds of transport processes, projected on a vertical axis. In particular it also represents mixing along inclined isopycnal surfaces.
The box-diffusion model is calibrated using the natural 14C distribution in the ocean and/or the penetration of bomb-produced 14C. It can be validated against two observed carbon cycle perturbations:
(a) the decrease of atmospheric 14C/ 12C due to fossil CO2 (Suess effect)
(b) the natural 14C variations as observed in tree-rings.
Figure 15.5 D14C of atmospheric CO2 (left-hand scale) and in ocean surface water (right-hand scale) in response to the input of bombproduced 14C. After Broecker and Peng (1982)
Figure 15.6 Four-reservoir model of CO2 exchange consisting of atmosphere and mixed layer (well mixed boxes), and the long-term biosphere and an eddy diffusive deep sea. Oeschger et al. (1975)
The atmospheric 14C/ 12C ratio decreases due to the emission of 14C-free fossil CO2; the natural 14C variations are most probably caused by fluctuations of the 14C production which can be derived from the 10Be deposition history in polar ice cores.
(a) Suess effect: The BD model calibrated with bomb-produced 14C predicts a 14C/ 12C decrease of
1.8% (Siegenthaler, 1983), while best observational results, taking into account 14C production variations due to changing solar modulation, lead to a value of
1.7% (Stuiver and Quay, 1981).
(b) Recent AMS measurements [Beer et al. , 1983] of the cosmic ray produced isotope 10Be in an ice core from Milcent, Greenland, have provided an approximate record of the radioisotope production variations in the
atmosphere due to solar modulation. 10Be, unlike 14C, is attached after production to aerosols and in a few months to about
1 year deposited with precipitation on the surface. Polar ice cores therefore provide a continuous record of the 10Be content of snowfalls, reflecting production of this radioisotope by cosmic radiation.
Figure 15.7 (top) shows the observed 10Be concentrations for the period 1200 to
1800 AD. A strong increase is observed for the period of the quiet sun (1640 to 1710), indicating reduced shielding due to solar magnetism. The 14C variations during the corresponding period
(Figure 15.7, bottom) show quasi-periodic variations with a period of ca. 200 years. Comparison of the 14C variations observed in tree-rings (dashed curve in
Figure 15.7, bottom)
with those calculated by the BD-model (Figure 15.7, bottom, solid line), assuming that the 14C production variations are proportional to those of 10Be, shows a satisfactory agreement. This demonstrates that the 10Be and the 14C variations are caused largely by the same phenomenon, i.e. solar modulation of cosmic radiation and that the BD-model is capable of providing correct attenuations and phase shifts for perturbations of this type. However, it needs to be emphasized that the BD-model is a simple simulation model and that the development of physical ocean-circulation models incorporating the carbon cycle is highly desirable. An interesting approach is the model by Maier- Reimer and
Hasselmann, 1987.
Figure 15.7 Top: 10Be concentration in units of 104 atoms/g ice as a function of age in the Milcent ice core , (Greenland). The age was determined based on annual variations of d18O. Bottom: 14C in the atmosphere. Dashed curve: tree-ring measurements; solid curve: 14C model-calculated based on production rates determined from the 10Be data. Beer et at. (1983)
The anthropogenic CO2 increase and the deconvolution of the CO2 input history using an ocean CO2 uptake model
Next we discuss the anthropogenic CO2 input due to deforestation and during the last decades mainly due to fossil fuel consumption. Precise continuous measurements of the atmospheric CO2 content have only been performed since 1958 on Mauna Loa, Hawaii, and at South Pole Station (Keeling et al. , 1982). Data prior to 1958 are aft1icted with uncertainties due to a variety of reasons. As mentioned before, the bubbles in polar ice constitute physically occluded samples of ancient air. During the last decade the analysis of the air bubbles in ice cores has been perfected, and information on the air occlusion mechanism has been gained. At Siple Station, Antarctica, in a joint US-Swiss operation, an ice core has been obtained which enabled an astonishingly precise reconstruction of the atmospheric CO2 increase (Neftel et al. , 1985) since the first half of the last century (Figure 15.8).
These results indicate a preindustrial atmospheric CO2 concentration near 280 ppm before about 1800 AD. Raynaud and Barnola, 1985, quote a preindustrial value of 260 ppm. Reasons for the discrepancy are being investigated. Measurements on ice cores from the South Pole support our preindustrial value and indicate that during the past preindustrial millenium the atmospheric CO2 concentration showed only minor changes ( < 10 ppm).
Figure 15.8 Measured mean CO2 concentration plotted against the estimated mean gas age. The horizontal axis of the ellipses indicates the close-off time interval of 22 y. The uncertainties of the concentration measurements are twice the standard deviation of the mean value. Neftel et al. (1985)
The increase, as shown in Figure 15.8, can therefore be unambiguously attributed to human activities. It started in the first half of the last century, probably mainly due to deforestation. In the middle of the 20th century the input due to fossil fuel consumption became dominant.
In the recent past it has also been possible to obtain d13C data on CO2 extracted from the Siple ice core. These data will add to the establishment of the anthropogenic CO2 input history and the disentangling of the different sources.
Taking all these results together, one gets the impression that a more and more consistent picture of the human impact on the CO2 cycle is evolving: The atmospheric CO2 concentration has been increasing monotonously since the first half of the 19th century. The major sink of the emitted CO2 is the ocean, and we seem to know the response of the ocean system to a perturbation at the atmosphere/ocean interface sufficiently well to give a reasonable estimate of the anthropogenic CO2 production function.
Of course, all the elements needed to assess in detail the human impact on the carbon cycle, i.e. the CO2/air, 13C, and 14C records, the CO2 uptake by the ocean and possibly by the biosphere and so forth, are still afflicted with uncertainties, and further strong research efforts are required.
The long-term 14C variations: trend in ocean dynamics or in 14C production?
The 14C record in tree-rings not only shows short-term fluctuations as discussed earlier, but also a long-term trend. In Figure 15.9 we see that 9000 to 10000 years ago the atmospheric 14C/ 12C ratio was 8 to 10% higher than before nuclear weapon testing started in the early 1950s (Suess, 1970). The question is often posed, is the decrease from higher 14C/ 12C ratios at the end of the last glaciation to lower ones during the last 2000 years a result of a change in production, e.g. due to an increasing geomagnetic shielding of cosmic radiation, or does it reflect a speeding up of ocean circulation and mixing which brings the atmospheric and mixed layer 14C/ 12C ratios closer to that of the subsurface ocean?
In our natural archives we can find in principle the answers to this question. The first test is to follow the 10Be profile in ice cores back into the last glacial period (Figure 15.10). An interesting result is the absence of an increase until about 5000 years before present. Further back, when approaching the glacial-postglacial transition, higher 10Be concentrations can indeed be observed. However, in this time range a different climatic regime is approached. Lower precipitation rates lead to a reduced dilution of atmospheric constituents and therefore higher 10Be concentrations. Thus it is difficult to reconstruct the 10Be fluxes at the glacial-postglacial transition and to normalize them to present conditions.
Figure 15.9 14C measurements on tree-rings (Bristlecone Pine), corrected for the radioactive decay. Deviations from a standard are given in permil. Neftel et al. (1981)
Figure 15.10 Upper part: 10Be concentration measured on the Camp Century ice core plotted versus time. Lower part: comparison between 14C data (dotted line) calculated with the carbon cycle model using the 10Be concentrations and the 14C data (solid line) measured on tree-rings. Beer et al. (1988)
14C measurements on foraminifera in ocean sediments offer another possibility to answer the question raised above. The 14C/12C ratio in surface water is reflected in shells of planktonic (ocean surface dwelling) foraminifera, and that of bottom water in shells of benthonic (ocean bottom dwelling) foraminifera. In high accumulation deep sea cores, where bioturbation only slightly affects stratigraphy, samples of coexisting foraminifera of the two types can be collected for 14C analysis.
This experiment has been successfully performed on a sediment core from the South China Sea, and interestingly enough the apparent age difference between the two foraminifera species remained, within the error limits, essentially constant (Andree et al. , 1986). At least, based on the information from this part of the ocean, no major change in ocean circulation and mixing is indicated in the Holocene. If this finding would hold for the entire ocean, the long-term trend in atmospheric 14C could not be explained by a shift in the ocean dynamics but rather in higher 14C production at the end of the last glaciation and the glacial-postglacial transition.
The climate system
The average global energy balance at the earth's surface gives indications regarding the most important parameters and mechanisms governing the earth's climate. It can be expressed as follows:
SpR2(1-A) = 4pR2sT4s(1 -B)
with S = solar constant
R = radius of the earth
A = albedo (reflected fraction of solar irradiation)
s = Stefan-Boltzmann constant
B = fraction of infrared radiation (emitted from surface), absorbed in atmosphere, and re-emitted back to surface
Ts = surface
temperature
The ocean also plays an important role regarding storage and distribution of heat in interaction with atmospheric circulation. Variations of the parameters S, A, and B lead to changes in the passive parameter Ts, the earth's surface temperature. Information on all these four parameters are recorded in natural archives.
The varying emission of solar plasma leads to changes in the magnetic shielding of galactic cosmic radiation in the inner part of the solar system. This produces variations in the cosmic ray flux reaching the earth. They are reflected as variations in the production of radioactive nuclei in the earth's atmosphere, as discussed above. The changes in the production rate are recorded in tree-rings as 14C/12C ratio changes and in precipitation as changes in the concentrations of the cosmogenic radio-isotopes 10Be and 36Cl. Solar properties like luminosity and ultraviolet emission might also influence climate. If they are related to the solar plasma emission-which seems to be plausible-changes in the 10Be content in ancient deposited snowfalls therefore reflect changes in solar parameters (like S) which influence climate.
Atmospheric turbidity alters the earth surface's albedo (A ). It is influenced by volcanic eruptions which lead to stratospheric dust layers. Solid electrical conductivity measurements on ice cores enable the identification of volcanic dust layers and therefore contribute to the reconstruction of the history of atmospheric turbidity.
During periods of glaciation, due to the enhanced continental and sea-ice cover, the albedo (A) is significantly increased. The 18O/16O ratio in the continental ice is considerably depleted compared to the ocean water. This leads to small enrichments in the oceanic 18O/16O ratio during glaciations which are reflected in the carbonate shells of foraminifera deposited in ocean sediments and enable, therefore, to estimate the history of the ice-cover of the continents.
As mentioned before, in natural ice essentially undisturbed air samples from the ancient atmosphere are stored. Measurements of the gas composition reveal variations of the contents of infrared active gases like CO2 and CH4, which influence the parameter B.
Variations in the 18O/160 ratio in precipitations reflect changes in temperature ( Ts ). Relative to sea-water, in cold periods precipitations are strongly depleted in 18O, while in warm periods the depletion is less. Ancient precipitation is stored in cold glacier ice. Under favourable conditions, the high resolution of isotopic information in ice cores enables even the reconstruction of seasonal 18O variations as far back as 10000 years.
Climatic change of the last 100 to 1000 years
Unlike the case of CO2 increase and its interpretation, the global climatic trend of the last 100 years, as estimated in Figure 15.11, cannot yet be unambiguously attributed to changes of the parameters S, A, and B. There exist different models which attempt to reconstruct a possible change in solar luminosity. Precise measurements of S from satellites might help to identify trends, perhaps correlated with solar activity and with radioisotope production. Attempts to reconstruct the history of the albedo A often make use of information on the atmospheric turbidity, based on the sulphate content recorded in Greenland ice cores which may or may not be a valuable measure of global albedo variations.
Figure 15.11 Surface air temperature changes representative of the Northerm Hemisphere land masses from Vinnikov et al. (1980). Hansen et al. (1981), and Jones et al. (1982). Smooth curves were obtained by using a 10-year Gaussian filter. Figure and caption from Wigley et al. (1985)
It would be most valuable if, in addition to these parameters determining the globally averaged energy balance of the earth, information regarding changes and anomalies in atmospheric and oceanic circulation could also be obtained. As mentioned before, the El Niño phenomenon is connected with considerable anomalies of the Pacific Ocean/atmosphere coupling. As an example, in December 1982, in parts of the Pacific Equatorial Ocean, the sea surface temperature had been ca. 3 to 5 °c above the average. It should be possible to reconstruct such anomalies from the 18O/16O ratios in low latitude glaciers and possibly in other natural archives like tree-rings. Alpine type and polar glaciers should also enable the reconstruction of atmosphere/ocean coupling anomalies, based on the deposition history of continental dust.
As Figure 15.12 indicates, the 1982-83 El Niño event also showed an effect on the global atmospheric CO2 content. If such a change of the order of 1 ppm yr-1 had lasted over a longer period of time, it would have led to a change in the atmospheric CO2 content of such a size that it should be detectable by very precise CO2 measurements on ice cores. Parallel 13C/12C measurements would enable us to distinguish between CO2 changes due to varying ocean surface properties and those due to varying terrestrial biomass. The precision of CO2 concentration measurements on ice cores has by now been improved to ca. ± 1.5 ppm, and CO2 concentration shifts of a few ppm during periods of a somewhat different climate, like the Medieval Warm , period or the Little Ice Age, would indicate a possible oceanic climatic change component.
Figure 15.12 Year-to-year global atmospheric CO2 change by season, plotted in the forward year. Data are from the 24-station global flask network of NOAA/GMCC [Komhyr et al., 1985; R. H. Gammon, NOAA, personal communication, 1984]. Seasonal global means are weighted. Figure and caption from Gammon et al., 1985
Long-term climatic change
Variation of the orbital parameters of the earth and the glacial cycles
Even for constant solar luminosity the intensity of solar radiation reaching the earth's surface and its latitudinal distribution undergo changes due to variations in the geometry of the earth's orbit around the sun. These changes, a result of the gravitational interaction in the planetary system, involve the eccentricity of the orbit and the obliquity and the precession of the earth's axis. They produce significant changes in the pattern of the incoming radiation on time-scales from several thousand to several hundred thousand years. These changes can be extrapolated many hundred thousand years into the past and can also be calculated into the future. Milankovitch (1930) suggested that these variations have caused the glacial periods. The averaged irradiation over the globe is not much changed by these orbital variations. The important effect is the modulation of the seasonal cycle. Summer insolation varies by as much as ±5% at latitudes at which continental ice existed. A comparison of the northern summer insolation history with the ice mass record, reflected in the foraminifera of deep-sea cores, shows that periods of rapid ice disappearance coincide with peaks of higher summer insolation. Furthermore, frequency analyses of the sedimentary oxygen isotope record reveal the characteristic frequencies of the orbital changes. It is widely accepted today that the orbital element changes play a major role in controlling long-term climate.
The modulation of the earth's climate by changes in the orbital parameters can also be seen in the d18O record of an Antarctic ice core from Vostok Station. (Lorius et al., 1985). In contrast to this Antarctic d18O profile, spanning the last 150 000 yr, in the Greenland ice cores the orbital effect is much less pronounced, and short term climatic variations there seem to play an important role.
Short-term variations: Information from Greenland ice cores and lake sediments
Superimposed on the long-term glacial cycles, there are rather drastic short- term climatic changes. An example is the so-called Little Ice Age in the period from the middle of the 16th to the middle of the 19th century, followed by a significant temperature increase, leading, for example, in Central Europe to a strong retreat of the Alpine glaciers. Information on short-term climatic changes is not available from deep sea sediment cores: bioturbation leads to a continuous mixing of the uppermost sediment layers, and the low accumulation rates do not allow a sufficient time resolution. More detailed climatic information is stored in ice cores and lake sediments. In Figure 15.13 the d18O recores of the recently recovered Dye 3 core are compared with that of the Camp Century core (Dansgaard et al., 1982). The good correlation between the two d18O records is clearly visible. This not only holds for the glacial-postglacial transition 10000 years ago but also for most of the pronounced d18O oscillations in the ice from the Wisconsin stage. The shifts of d18O at the transition to the Holocene 13 000 and 10 000 years BP are also recorded in carbonate deposits of Central European lakes. Figure 15.14 demonstrates the excellent correlation. The rapid d18O oscillations reflect relatively warm periods in the generally cold Wisconsin stage. Some of them may have their counterparts in pollen profiles of peat bogs or lake sediments from Central Europe. Such pronounced correlations are not found in climatic records from the North American continent. Ruddiman and McIntyre (1981) found evidence in North Atlantic sediment cores for changes of the North Atlantic polar front (Figure 15.15), leading to deviations of the Gulf Stream which then caused the climatic variations in Central Europe.
Figure 15.13 d18O profiles measured in Copenhagen along the Dye 3 (0 to 1982 m depth) and the Camp Century (0 to 1370 m depth) ice cores plotted on a common linear time-scale based on considerations discussed by Dansgaard et al. (1982)
Figure 15.14 Comparison of a section of the d18O profile from the Dye 3 ice core (right) with the d18O record in lake Gerzensee (left). The strong similarities suggest that both records represent the same sequence of climatic events and thus the same time period. Oeschger et al. (1984)
Figure 15.15 Location of the North Atlantic polar front for different time periods [Ruddiman and McIntyre, 1981]. Deglacial retreat was interrupted by the readvance from 11-10 kyr
Ice core parameters during the Wisconsin and the Wisconsin/Holcene transition
In the discussion of experimental evidence on the carbon cycle, we mentioned that during the last thousand years the atmospheric CO2 concentration had been essentially constant in the range of 280 ±5 ppm. But does this hold for periods of major climatic change? It is one of the most surprising observations on ice cores that at the end of the Wisconsin the CO2 concentration ranged between 180-200 ppm and then increased parallel to the d18O shift to values of 260 to 300 ppm (Stauffer et al. , 1984). The CO2 measurements on the recently drilled Dye 3 ice core confirm this observation (Figure 15.16). A rapid transition in both d18O and CO2 is observed around 13000 BP. The CO2 transition seems to lead the d18O transition in the ice core by a few metres. This depth difference might be explained by the fact that the gases get occluded in cold ice at a depth of ca. 100 ± 30 m. The phase shift might therefore reflect the original trapping depth of air, and it cannot be excluded that the parameters CO2/air and d18O changed simultaneously. A high CO2 concentration is also found in the Dye 3 ice at a depth of 1890 m, corresponding to an age of approximately 40 000 BP. This value coincides with one of the high 180 periods during the Wisconsin. Figure 15.17 shows the result of a detailed study of the d18O/CO2 relationship in the 30000 to 40000 year old section of the Dye 3 core. All the rapid d18O oscillations are accompanied by simultaneous, perfectly correlated CO2 oscillations. Though the observation of rapid atmospheric CO2 concentration changes during the Wisconsin needs further confirmation by measurements on other ice cores to exclude artefacts due to melt-layers or interaction with the impurities in the ice lattice, these experimental results have inspired the discussion of mechanisms which might produce rapid atmospheric CO2 changes of the observed extent.
Figure 15.16 From top to bottom: 10Be concentration, CO2 concentration (ppm) and d18O data obtained for the Dye 3 ice core. The tentative time marks are suggested by the comparison with 14C dated European lake sediments (see Figure 15.14)
Other parameters vary in a similar way over this time interval. Microparticle concentrations show strong d18O correlated variations, concentrations being more than a factor of six higher during cold periods than during warm ones. The cold/warm ratios are about 1.5 for chlorine and nitrate and about 4 for sulphate (Finkel and Langway, 1985). Because of the larger sample requirements for the 10Be analyses, measurements could not be made with the same resolution as for the other species, but in core sections with high d18O, the 10Be values are also 2 to 2.5 times higher than compared to low d18O periods.
Figure 15.17 CO2 and d18O values measured on ice samples from Dye 3 (the 30 m increment corresponds to about 10000 years). a) Circles indicate the results of single measurements of the CO2 concentration of air extracted from ice samples. The solid line connects the mean values for each depth. b) The solid line connects the d18O measurements done on 0.1 m core increments. Stauffer et al. (1984)
It is difficult to interpret these phenomena in detail. The relative concentration shifts are not the same for all species. Therefore, they cannot only be the result of a simple dilution modulation of a constant impurity flux by a variable water flux system.
The bistable climatic system
Not much is known for sure about the mechanisms determining the state and the stability of the climate system. The most remarkable observation in the data set from the Dye 3 core is the apparent bistable nature of the observed variations. Cold and warm period values of all parameters tend to occur in bands the width of which is significantly smaller than the difference between them. The correlated variation of the parameter sets suggests the existence of a bistable climate system during the Wisconsin, one set describing the warm and the other the cold climate state.
The rapid climatic oscillations during the Wisconsin are probably internally forced changes of the climate system. During the transition times, the essential external parameters and boundary conditions like insolation pattern, sea-level, and continental ice-cover probably changed in a very limited range. It seems therefore possible that for a given set of boundary conditions the atmosphere-ocean system has more than one mode of operation. After a period of one to two thousand years, the stabilizing mechanisms of one mode of operation seem to become ineffective, and the system organizes itself in a new mode. Based on the Greenland ice core records and European climatic records, one gets the impression that during the ice age essentially two separated climatic modes existed. About 10000 BP boundary conditions as high latitude summer insolation seem to have prevented the system from switching once more back into the cold state. Based on European continental climatic information, we know that around 13 000 BP almost Holocene climatic conditions existed. Around 11 000 BP the system switched back for the last time, until present, to the cold mode, leading to almost glacial conditions in Europe. After the transition to the warm state around 10000 BP, the stability was already so high that a return to the cold state was impossible. Since then the system has remained essentially in the warm state, and it is important to note that during the Holocene, the climate, as reconstructed by d18O, has been much more quiet than during the Wisconsin. It seems that the large amounts of continental ice played an important role in the pronounced variability of the climate system during the last glaciation. Broecker et al. (1985) suggested that the rapid Wisconsin climatic variations reflect switches of deep water formation between the North Atlantic and the North Pacific Ocean.
The ocean and atmospheric CO2 changes
At present, the low atmospheric CO2 concentrations at the end of the last glaciation have been observed in three ice cores from Antarctica and two ice cores from Greenland. It is therefore highly probable that the atmospheric CO2 content did indeed show these fluctuations. Measurements on the Vostok ice core, Antarctica, even show a similar CO2 shift from low 'Riss' glaciation CO2 concentration to Eemian interglacial CO2 concentration in the range of those observed for the Holocene. The CO2 changes therefore seem to be an inherent mechanism of the glacial cycle.
Regarding the rapid CO2 changes, observed in the Greenland cores during the Wisconsin, however, the final proof that they reflect atmospheric changes could not yet be given.
As was indicated earlier, the atmospheric CO2 concentration depends on the chemical composition of the ocean surface water which in turn is strongly influenced by marine biological activity. Broecker (1982) suggested that the observed CO2 content changes must be the result of alterations in the nutrient element chemistry of sea-water. A higher phosphate content in the glacial ocean would have led to a stronger depletion of total CO2, and thus pCO2 in, the surface ocean. In one of his models he suggested that the source of this additional phosphate was erosion of nutrient-rich organic sediments on the continental shelves exposed due to the lower sea-level in glacial times. This hypothesis, though extremely important for the initiation of attempts to explain the observed CO2 changes, now seems to disagree with the observations which ask for rather rapid CO2 changes. Today, therefore, the reason for the CO2 changes is searched for in changes of the highly interactive system of physical, chemical, and biological processes in the ocean system. In the Southern Ocean around Antarctica, the nutrients phosphate and nitrate are abundant in surface waters in contrast to most of the other part of the ocean. The biological activity seems to be limited around Antarctica due to rapid vertical mixing or lack of solar light. Several authors showed that a decrease of the vertical mixing in the Southern Ocean, assuming the biological productivity to remain constant, would lead to a lower surface concentration of nutrients, total CO2 and consequently pCO2. Changes in atmospheric CO2 by 50 ppm within a few 100 years could be produced by halfing the surface/deep water exchange rate.
It was previously mentioned that the average difference of d13C in surface and deep ocean water is a measure for the ocean's average biological activity. The time history of d13C in surface and deep ocean water is recorded in coexisting planktonic and benthic foraminifera in ocean sediment samples. Shackleton and Pisias (1985) provided for a Pacific sediment core a rec6rd of the ice volume based on d18O measurements and of the difference of the d13Cratios in surface and deep ocean dwelling foraminifera. They assumed the d13C difference to be representative for the average oceanic biological activity and derived a record of the atmospheric CO2 content of the last 350 000 years (Figure 15.18). Their data confirm the low CO2 concentrations at the end of the last glaciation as measured in ice cores. A spectral analysis of the CO2 variation showed significant amplitudes for the Milankovitch frequencies of the changing orbital parameters. These studies show that both CO2 and ice volume lag the orbital changes. And there is most exciting evidence-which should be confirmed by additional measurements-that the changes of CO2 seem to lead those in ice volume by a few thousand years.
Figure 15.18 Surface-to-deep water 13C difference. Scales are shown (1) in permil (by subtraction); (2) in parts per million by volume atmospheric CO2 concentration implied by scale 1; (3) in mean phosphorus content of ocean deep water that would be required to produce scale 1; (4) in mean Redfield ratio that would be required to explain scale 1(a) (note that scales 3 and 4 are presented as alternatives) Shackleton and Pisias (1985)
The ocean's role in the anticipated global change
The short-term reaction of the atmosphere/ocean CO2 system to the anthropogenic CO2 immission is rather well known. The preindustrial atmospheric CO2 concentration (as well as that of other trace gases) can be reconstructed within narrow uncertainty limits. Using the reconstructed and measured atmospheric CO2 increase and models of the CO2 uptake by the oceans, the CO2 immission originating from deforestation and from fossil fuel consumption can be calculated, and a satisfactory agreement with independent production estimates is obtained.
The rising concentrations of CO2 and other trace gases are estimated to have already led to a significant global temperature increase. However, at present it is not possible to deduce in a scientifically rigorous manner to what degree the global temperature increase of ca. 0.5 °c during the last hundred years results from an increasing greenhouse effect and to what extent from other factors, like changing solar parameters, atmospheric turbidity or oceanic behaviour .
There exists considerable uncertainty regarding the long-time role of the ocean in global change. The expected temperature increase will lead to density changes of surface ocean water due to thermal expansion, influx of fresh water from melting continental ice but also due to changes in the evaporation -precipitation balance. These potential changes in the ocean behaviour seem to be the greatest uncertainty in the prediction of regional climatic change. But a changing ocean might also affect the future trend of the atmospheric CO2 concentration and the global temperature increase:
-A slowing down of oceanic circulation and mixing, due to a reduced latitudinal temperature gradient and reduced ocean surface water density, might reduce the ocean's uptake of excess CO2 and its transport to greater depths.
-A changing oceanic circulation pattern might also influence the baseline CO2 concentration as discussed earlier
-The dampening of the anticipated temperature rise due to the ocean's large heat capacity would be reduced if vertical ocean mixing slowed down.
The ocean/CO2 and ocean/climate coupling at present and in the past
The interaction between ocean, CO2, and climate is clearly demonstrated by the El Niño events which repeat themselves within 3 to 7 years and lead to significant climatic anomalies as well as small, but clearly identifiable, irregularities in the atmospheric CO2 increase. In 1982 these phenomena were especially pronounced, and sometimes the question is posed, will the anticipated global change manifest itself in more prominent anomalies of ocean circulation.
An example of the strong influence of ocean circulation on climate (mainly on the European continent) are the events at the glacial-postglacial transition reconstructed from palaeoclimatic information. A strong link between changes in the North Atlantic Ocean circulation and rapid changes of the European climate have been observed. The rapid changes of d18O and other parameters in the Greenland ice cores further suggest the existence of a bistable climatic system North Atlantic-Europe.
The observation that climatic events are accompanied by changes in the atmospheric CO2 concentration is of great interest for two reasons:
(a) The major climatic shifts over the last 150000 years are strongly correlated with shifts in the atmospheric CO2 concentrations. During the coldest periods of the two last glaciations (18000 BP and 140000 BP) the CO2 concentrations were of the order of 180-200 ppmv. The CO2 concentrations rose almost simultaneously with the climate shifts to values of 270-290 ppmv in the subsequent interglacial. Thus CO2 shifts by a factor of about 1.4 seem to be inherent to major climatic variations. The CO2 shifts between glacial and interglacial stages are probably mainly forced by changes in oceanic
circulation and/or marine biological activity.
(b) The link between ocean circulation and CO2 anomalies perhaps offers the possibility to trace back changes in oceans by very precise high resolution measurements of CO2 concentrations of the air occluded in polar ice cores. With the precision achieved at present, it should be possible to detect atmospheric CO2 concentration changes of
³ 2 to 3 ppm over periods of ³
20 years. This might provide otherwise not obtainable information on oceanic circulation changes which could then be linked to climatic deviations like the Mediaeval Warm Period and the Little Ice Age in Europe. Such information would
significantly help us to understand major climatic mechanisms involving the ocean.
The importance of improving the knowledge of ocean processes relevant to climate and CO2 has been widely recognized by the scientific community. This is exemplified by a series of international scientific projects related to questions raised in this chapter which are coordinated by the Committee on Climatic Changes and the Ocean (CCCO):
The World Ocean Circulation Experiment (WOCE) is concerned with improving our understanding of ocean circulation mechanisms and the ability to model global circulation.
The tropical Ocean-Global Atmosphere Project (TOGA) is focused on the attempt to understand and describe the tropical oceans and the global atmosphere as a coupled system for the purpose of predicting its variations on time-scales of months to years.
A new programme is being planned by CCCO to Improve the Understanding of the Carbon Cycle in the Ocean. The main aspects are:
(a) Measurements over time of the main constituents of the CO2 system in surface and subsurface ocean waters and the rates and locations of CO2 exchange between air and sea
(b) studies of the interaction between biological activity in the sea and atmospheric and oceanic CO2, including determinations of organic particle fluxes from the euphotic zone into deeper waters and the chemical transformations of the particles. The physical and chemical measurements planned in WOCE and TOGA should be extended to improve predictions of the response of the carbon cycle to changing climate during the next century.
Another activity in the frame of CCCO is palaeoclimatology. Considerable progress has been made in this field, as discussed in this article, due to the availability of new analytical methods (e.g. accelerator mass spectrometry) and the application of fundamental principles of geochemistry to the reconstruction of past climate and oceanic properties. Studies of primary importance comprise reconstructions of variations of the climate and the CO2 systems, process studies and model experiments involving the biochemistry and the dynamics of the ocean, with special emphasis on the past abrupt climatic changes.
Efforts should particularly concentrate on an observational programme to obtain time series of environmental parameters suitable to identify important mechanisms and to validate models. The needs for new deep ice cores, new high resolution deep sea sediment records, and lake and other continental records are stressed.
In the following we want to expand on the importance of palaeoclimatological studies and propose a systematic coordinated observational programme together with modelling attempts of the ocean/climate/CO2 interaction.
In the next few decades a considerable part of the progress in our understanding of the ocean system and its role in environmental processes will be due to information on past variations in ocean properties in relation with other changes in the entire environmental system. Therefore a new project should be initiated which coordinates and reinforces palaeoclimatic research, making use of state of the art analytical techniques to study the information contained in both oceanic and continental records. This project should be aimed at providing:
-links between oceanic and continental events,
-a basis for the development and validation of a wide hierarchy of models involving the coupled
atmosphere-ocean-kryosphere system including biological processes.
Such a project could be called Palaeoenvironmental Traverse. Time periods of major interest are:
-1950 until present: Period of detailed instrument data on ocean and atmosphere with anomalies in ocean circulation.
-1850-1950: Period of rising human impact on the environment (CO2 emission, consequences of population increase, changes in atmospheric
composition).
-Last 1000 years: Period of significant climatic deviations like the Mediaeval Warm and the Little Ice Age in Europe.
-Altithermal (6000 to 8000 BP): Period of warmer earth similar to that anticipated for the first half of the next century due to human impacts.
-Glacial-Postglacial Transition (15000 to 9000 BP): Period of pronounced climatic events involving ocean and
kryosphere.
-Wisconsin: Period of rapid climatic oscillation between a cold and a warm climatic system state in the North Atlantic region.
Andree, M., Oeschger, H., Broecker, W., Beavan, N., Klas, M., Mix, A., Bonani, G., Hofmann, H. J., Suter, M., Wolfli, W., and Peng, T.-H. (1986). Limits on the ventilation rate for the deep ocean over the last 12000 years. Climate Dynamics, 1, 53-62.
Beer, J., Siegenthaler, U., Oeschger, H., Andree M., Bonani, G., Suter, M., Wolfli, W., Finkel, R., and Langway, C. C. (1983). Temporal 10Be variations. Proc. 18th Int. Cosmic Ray Conf., Bangalore, 9, 317.
Beer, J., Siegenthaler, V., Bonani, G., Finkel, R. C., Oeschger, H., Sutter, H. and Wolfli, W. (1988). Information on past solar activity and geomagnetism from 10Be in the Camp Century ice core. Nature (in press).
Broecker, W. S. (1982). Ocean chemistry during glacial time. Geochimica et Cosmo- chimica Acta, 46, 1689-1705.
Broecker, W. S., and Peng, T.-H. (1982). Tracers in the Sea. Eldigio Press, Lamont-Doherty Geological Observatory, Palisades, NY 10964: 690 pages.
Broecker, W. S., Peng, T.-H., and Engh, R. (1980). Modelling the carbon system Radiocarbon, 22, 565-598.
Broecker, W. S., Peteet, D. M., and Rind, D. (1985). Does the ocean-atmosphere system have more than one stable mode of operation? Nature, 315, 21-26.
Dansgaard, W., Clausen, H. B., Gundestrup, G., Hammer, C. U., Johnsen, S. F., Kristinsdottir, P. M., and Reeh, N. (1982). A new Greenland deep ice core. Science, 218, 1273-1277.
Finkel, R. C., and Langway, C. C., Jr. (1985). Global and local influences on the chemical composition of snowfall at Dye 3, Greenland: the record between 10 ka BP and 40 ka BP. Earth and Planetary Science Letters, 73, 196-206.
Gammon, R. H., Sundquist, E. T. , and Fraser, P. J. (1985). History of carbon dioxide in the atmosphere. In Trabalka, J. R. (Ed.) Atmospheric Carbon Dioxide and the Global Carbon Cycle, pp. 25-62. U.S. Department of Energy, Washington, DC.
Hansen, J., Johnson, D., Lacis, A., Lebedeff, S., Lee, P., Rind, D., and Russell, G. (1981). Climatic impact of increasing atmospheric carbon dioxide. Science, 213, 957-966.
Jones, P. D., Wigley, T. M. C., and Kelly, P. M. (1982). Variations in surface air temperatures: Part I. Northern Hemisphere 1881-1980. Monthly Weather Review, 110, 59-70.
Keeling, C. D., Bacastow, R. B., and Whorf, T. P. (1982). Measurements of the concentration of carbon dioxide at Mauna Loa Observatory, Hawaii. In Clark, W. C. (Ed.) Carbon Dioxide Review, 1982, 377-385. Oxford University Press, New York.
Komhyr, W. D., Gammon, R. H., Harris, T. B., Waterman, L. S., Conway, T. J., Taylor, W. R., and Thoring, K. W. (1985). Global atmospheric CO2 distribution and variations from 1968-1982. NOAA/GMCC Flask Sample Data. J. Geophys. Res., 90, 5567-5596.
Lorius, C., Jouzel, J., Ritz, C., Merlivat, L., Barkov, N. J., Korotkevitch, Y. S., and Kotlyakov, V. M. (1985). A 150 000-year climatic record from Antarctic ice. Nature, 316, 591-596.
Maier-Reimer, E., and Hasselmann, K. (1987). Transport and storage of CO2 in the ocean-an inorganic ocean-circulation carbon cycle model. Climate Dynamics, 2.
Milankovitch, M., (1930). Mathematische Klimalehre und astronomische Theorie der Klimaschwankungen. In Handbuch der Klimatologie, I (A), Koppen, W., and Geiger, R. (Eds.) Gebruder Borntrager, Berlin.
Neftel, A., Oeschger, H., and Suess, H. E. (1981). Secular non-random variations of cosmogenic carbon-14 in the terrestrial atmosphere. Earth Planet. Sci. Lett., 56, 127-147.
Neftel, A., Moor, E., Oeschger, H., and Stauffer, B. (1985). Evidence from polar ice cores for the increase in atmospheric CO2 in the past two centuries, Nature, 315, No. 6014, 45-47.
Nydal, R., and Lovseth, K. (1983). Tracing bomb 14C in the atmosphere 1962-1980. J. Geophys. Res., 88, 3621-3642.
Oeschger, H., Beer, J ., Siegenthaler, U ., Stauffer, B., Dansgaard, W., and Langway, C. C. (1984). Late-Glacial climate history from ice cores. In Hansen, J., and Takahashi, T. (Eds.) Climate Processes and Climate Sensitivity, pp. 299-306. Am. Geophys. Union.
Oeschger, H., Siegenthaler, U., Schotterer, U., and Gugelmann, A. (1975). A box diffusion model to study the carbon dioxide exchange in nature. Tellus, 27, 168-192.
Raynaund, D., and Barnola, J. M. (1985). An Antarctic ice core reveals atmospheric CO2 variations over the past few centuries. Nature, 315, 309-311.
Ruddiman, W. F., and McIntyre, A. (1981). The North Atlantic Ocean during the last glaciation. Palaeogeogr., Palaeoclimatol., Palaeoecol., 35, 145-214.
Shackleton, N. J., and Pisias, N. G. (1985). Atmospheric carbon dioxide, orbital forcing, and climate. In Sundquist, E. T., and Broecker, W. S. (Eds.) The Carbon Cycle and Atmospheric CO2: Natural Variations Archean to Present, pp. 303-317. Geophysical Monograph, 32, AGU Washington, DC.
Siegenthaler, U. (1983). Uptake of excess CO2 by an outcrop-diffusion model of the ocean. J. Geophy.s. Res. , 88, 3599-3608.
Stauffer, B., Hofer, H., Oeschger, H., Schwander, J., and Siegenthaler, U. (1984). Atmospheric CO2 concentration during the last glaciation. Annals of Glaciology, 5, 160-164.
Stuiver, M., and Quay, P. D. (1981). Atmospheric 14C changes resulting from fossil fuel CO2 release and cosmic ray flux variability. Earth and Planetary Science Letters, 53, 349-362.
Suess, H. E. (1970). Bristlecone-pine calibration of the radiocarbon time-scale 5200 BC to the present. In Olsson, I. U. (Ed.) Radiocarbon Variations and Absolute Chronology, pp. 303-311. John Wiley & Sons.
Vinnikov, K. Ya., Gruza, G. V., Zakharov, V. F., Krillov, A. A., Kovyneva, N. P., and Rarikova, E. Ya, (1980). Current climatic changes in the Northern Hemisphere. Meteorologiya i Gidrologya, 6, 5-17.
Wigley, T. M. L., Angell, J. K., and Jones, P. D. (1985). Analysis of the temperature record. In McCracken, M. C., and Luther, F. M. (Eds.) Detecting the Climatic Effects of Increasing Carbon Dioxide, pp. 55-90. U.S. Department of Energy, Washington, DC.
|
|
|
The electronic version of this publication has been
prepared at |